NTA JEE Main 26th August 2021 Shift 2
For the following questions answer them individually
NTA JEE Main 26th August 2021 Shift 2 - Question 1
If the length of the pendulum in pendulum clock increases by 0.1%, then the error in time per day is:
NTA JEE Main 26th August 2021 Shift 2 - Question 2
The angle between vector $$\left(\vec{A}\right)$$ and $$\left(\vec{A} - \vec{B}\right)$$ is:
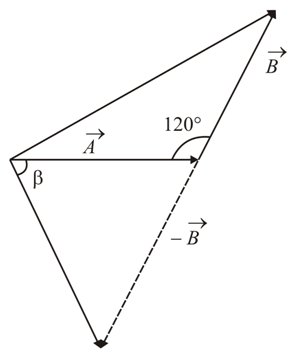
NTA JEE Main 26th August 2021 Shift 2 - Question 3
Match List - I with List - II:
List - I List - II
a. Magnetic induction i. $$ML^2T^{-2}A^{-1}$$
b. Magnetic flux ii. $$M^0L^{-1}A$$
c. Magnetic permeability iii. $$MT^{-2}A^{-1}$$
d. Magnetization iv. $$MLT^{-2}A^{-2}$$
Choose the most appropriate answer from the options given below:
NTA JEE Main 26th August 2021 Shift 2 - Question 4
A particle of mass $$m$$ is suspended from a ceiling through a string of length $$L$$. The particle moves in a horizontal circle of radius $$r$$ such that $$r = \frac{L}{\sqrt{2}}$$. The speed of particle will be:
NTA JEE Main 26th August 2021 Shift 2 - Question 5
A bomb is dropped by a fighter plane flying horizontally. To an observer sitting in the plane, the trajectory of the bomb is a:
NTA JEE Main 26th August 2021 Shift 2 - Question 6
The solid cylinder of length 80 cm and mass $$M$$ has a radius of 20 cm. Calculate the density of the material used if the moment of inertia of the cylinder about an axis $$CD$$ parallel to $$AB$$ as shown in figure is 2.7 kg m$$^2$$.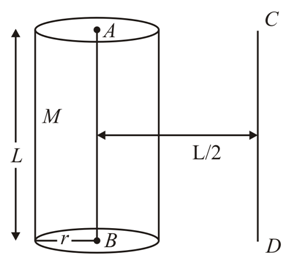
NTA JEE Main 26th August 2021 Shift 2 - Question 7
Two blocks of masses 3 kg and 5 kg are connected by a metal wire going over a smooth pulley. The breaking stress of the metal is $$\frac{24}{\pi} \times 10^2$$ N m$$^{-2}$$. What is the minimum radius of the wire?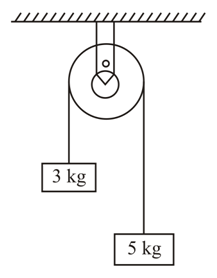
(take g = 10 m s$$^{-2}$$)
NTA JEE Main 26th August 2021 Shift 2 - Question 8
The temperature of equal masses of three different liquids $$x$$, $$y$$ and $$z$$ are 10°C, 20°C and 30°C respectively. The temperature of mixture when $$x$$ is mixed with $$y$$ is 16°C and that when $$y$$ is mixed with $$z$$ is 26°C. The temperature of mixture when $$x$$ and $$z$$ are mixed will be:
NTA JEE Main 26th August 2021 Shift 2 - Question 9
A refrigerator consumes an average 35 W power to operate between temperature -10°C to 25°C. If there is no loss of energy then how much average heat per second does it transfer?
NTA JEE Main 26th August 2021 Shift 2 - Question 10
A cylindrical container of volume $$4.0 \times 10^{-3}$$ m$$^3$$ contains one mole of hydrogen and two moles of carbon dioxide. Assume the temperature of the mixture is 400 K. The pressure of the mixture of gases is:
[Take gas constant as 8.3 J mol$$^{-1}$$ K$$^{-1}$$]


.webp)
