NTA JEE Main 25th July 2022 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 25th July 2022 Shift 2 - Chemistry - Question 31
The first ionization enthalpies of $$Be$$, $$B$$, $$N$$ and $$O$$ follow the order
NTA JEE Main 25th July 2022 Shift 2 - Chemistry - Question 32
Match List I with List II

Choose the correct answer from the options given below
NTA JEE Main 25th July 2022 Shift 2 - Chemistry - Question 33
$$K_{a1}$$, $$K_{a2}$$ and $$K_{a3}$$ are the respective ionization constants for the following reactions (a), (b) and (c).
(a) $$H_2C_2O_4 \rightleftharpoons H^+ + HC_2O_4^-$$
(b) $$HC_2O_4^- \rightleftharpoons H^+ + C_2O_4^{2-}$$
(c) $$H_2C_2O_4 \rightleftharpoons 2H^+ + C_2O_4^{2-}$$
The relationship between $$K_{a1}$$, $$K_{a2}$$ and $$K_{a3}$$ is given as
NTA JEE Main 25th July 2022 Shift 2 - Chemistry - Question 34
In base vs. Acid titration, at the end point methyl orange is present as
NTA JEE Main 25th July 2022 Shift 2 - Chemistry - Question 35
High purity ($$> 99.95\%$$) dihydrogen is obtained by
NTA JEE Main 25th July 2022 Shift 2 - Chemistry - Question 36
The correct order of density is
NTA JEE Main 25th July 2022 Shift 2 - Chemistry - Question 37
Arrange the following in decreasing acidic strength.
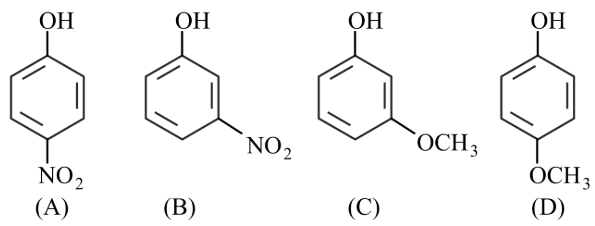
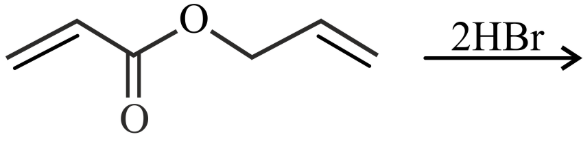
NTA JEE Main 25th July 2022 Shift 2 - Chemistry - Question 38
Major product of the following reaction is
NTA JEE Main 25th July 2022 Shift 2 - Chemistry - Question 39
Match List I with List II.

Choose the correct answer from the options given below
NTA JEE Main 25th July 2022 Shift 2 - Chemistry - Question 40
Two solutions A and B are prepared by dissolving $$1 \text{ g}$$ of non-volatile solutes X and Y respectively in $$1 \text{ kg}$$ of water. The ratio of depression in freezing points for A and B is found to be $$1:4$$. The ratio of molar masses of X and Y is
NTA JEE Main 25th July 2022 Shift 2 - Chemistry - Question 41
The molar conductivity of a conductivity cell filled with $$10$$ moles of $$20 \text{ mL NaCl}$$ solution is $$\Lambda_{m1}$$ and that of $$20$$ moles of another identical cell having $$80 \text{ mL NaCl}$$ solution is $$\Lambda_{m2}$$. The conductivities exhibited by these two cells are same. The relationship between $$\Lambda_{m2}$$ and $$\Lambda_{m1}$$ is
NTA JEE Main 25th July 2022 Shift 2 - Chemistry - Question 42
For micelle formation, which of the following statements are correct?
(A) Micelle formation is an exothermic process.
(B) Micelle formation is an endothermic process.
(C) The entropy change is positive.
(D) The entropy change is negative.
NTA JEE Main 25th July 2022 Shift 2 - Chemistry - Question 43
Given below are two statements.
Statement I: Pig iron is obtained by heating cast iron with scrap iron.
Statement II: Pig iron has a relatively lower carbon content than that of cast iron.
In the light of the above statements, choose the correct answer from the options given below.
NTA JEE Main 25th July 2022 Shift 2 - Chemistry - Question 44
The total number of acidic oxides from the following list is:
$$NO$$, $$N_2O$$, $$B_2O_3$$, $$N_2O_5$$, $$CO$$, $$SO_3$$, $$P_4O_{10}$$
NTA JEE Main 25th July 2022 Shift 2 - Chemistry - Question 45
The correct order of energy of absorption for the following metal complexes is
$$A: [Ni(en)_3]^{2+}$$, $$B: [Ni(NH_3)_6]^{2+}$$, $$C: [Ni(H_2O)_6]^{2+}$$
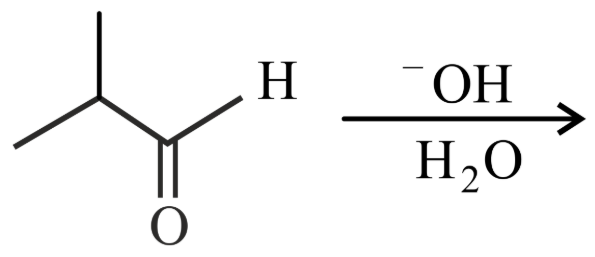
NTA JEE Main 25th July 2022 Shift 2 - Chemistry - Question 46
What is the major product of the following reaction?
NTA JEE Main 25th July 2022 Shift 2 - Chemistry - Question 47
The correct structure of C is

NTA JEE Main 25th July 2022 Shift 2 - Chemistry - Question 48
Match List-I with List-II

Choose the correct answer from the options given below
NTA JEE Main 25th July 2022 Shift 2 - Chemistry - Question 49
Some drugs bind to a site other than the active site of an enzyme. This site is known as
NTA JEE Main 25th July 2022 Shift 2 - Chemistry - Question 50
Glycosidic linkage between $$C_1$$ of $$\alpha$$-glucose and $$C_2$$ of $$\beta$$-fructose is found in
NTA JEE Main 25th July 2022 Shift 2 - Chemistry - Question 51
$$56.0 \text{ L}$$ of nitrogen gas is mixed with excess of hydrogen gas and it is found that $$20 \text{ L}$$ of ammonia gas is produced. The volume of unused nitrogen gas is found to be ______ L.
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2022 Shift 2 - Chemistry - Question 52
When the excited electron of a H atom from $$n = 5$$ drops to the ground state, the maximum number of emission lines observed are ______.
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2022 Shift 2 - Chemistry - Question 53
The sum of number of lone pairs of electrons present on the central atoms of $$XeO_3$$, $$XeOF_4$$ and $$XeF_6$$ is ______.
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2022 Shift 2 - Chemistry - Question 54
A sealed flask with a capacity of $$2 \text{ dm}^3$$ contains $$11 \text{ g}$$ of propane gas. The flask is so weak that it will burst if the pressure becomes $$2 \text{ MPa}$$. The minimum temperature at which the flask will burst is ______ °C. [Nearest integer]
(Given: $$R = 8.3 \text{ J K}^{-1} \text{ mol}^{-1}$$. Atomic masses of C and H are $$12u$$ and $$1u$$ respectively.) (Assume that propane behaves as an ideal gas.)
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2022 Shift 2 - Chemistry - Question 55
While performing a thermodynamics experiment, a student made the following observations:
$$HCl + NaOH \rightarrow NaCl + H_2O \quad \Delta H = -57.3 \text{ kJ mol}^{-1}$$
$$CH_3COOH + NaOH \rightarrow CH_3COONa + H_2O \quad \Delta H = -55.3 \text{ kJ mol}^{-1}$$
The enthalpy of ionization of $$CH_3COOH$$ as calculated by the student is ______ $$\text{kJ mol}^{-1}$$.
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2022 Shift 2 - Chemistry - Question 56
The separation of two coloured substances was done by paper chromatography. The distances travelled by solvent front, substance A and substance B from the base line are $$3.25 \text{ cm}$$, $$2.08 \text{ cm}$$ and $$1.05 \text{ cm}$$ respectively. The ratio of $$R_f$$ values of A to B is ______ (Answer the nearest integer).
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2022 Shift 2 - Chemistry - Question 57
The total number of monobromo derivatives formed by the alkanes with molecular formula $$C_5H_{12}$$ is ______ (excluding stereo isomers).
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2022 Shift 2 - Chemistry - Question 58
For the decomposition of azomethane $$CH_3N_2CH_3(g) \rightarrow CH_3CH_3(g) + N_2$$, a first order reaction, the variation in partial pressure with time at $$600 \text{ K}$$ is given as shown.
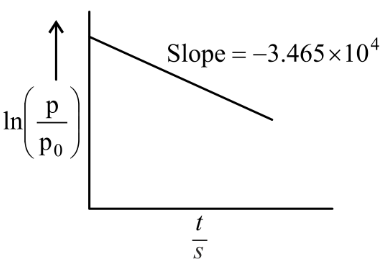
The half life of the reaction is ______ $$\times 10^{-5}$$ s.
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2022 Shift 2 - Chemistry - Question 59
The spin-only magnetic moment value of $$M^{3+}$$ ion (in gaseous state) from the pairs $$Cr^{3+}/Cr^{2+}$$, $$Mn^{3+}/Mn^{2+}$$, $$Fe^{3+}/Fe^{2+}$$ and $$Co^{3+}/Co^{2+}$$ that has negative standard electrode potential, is ______ B.M.
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2022 Shift 2 - Chemistry - Question 60
A sample of $$4.5 \text{ mg}$$ of an unknown monohydric alcohol, $$R-OH$$ was added to methylmagnesium iodide. A gas is evolved and is collected and its volume measured to be $$3.1 \text{ mL}$$. The molecular weight of the unknown alcohol is ______ g/mol.
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




