NTA JEE Main 25th July 2021 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Main 25th July 2021 Shift 1 - Chemistry - Question 31
The ionic radii of K$$^+$$, Na$$^+$$, Al$$^{3+}$$ and Mg$$^{2+}$$ are in the order:
NTA JEE Main 25th July 2021 Shift 1 - Chemistry - Question 32
At 298.2 K the relationship between enthalpy of bond dissociation (in kJ mol$$^{-1}$$) for hydrogen E$$_H$$ and its isotope, deuterium E$$_D$$, is best described by:
NTA JEE Main 25th July 2021 Shift 1 - Chemistry - Question 33
Given below are two statements:
Statement I : None of the alkaline earth metal hydroxides dissolve in alkali.
Statement II : Solubility of alkaline earth metal hydroxides in water increases down the group.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Main 25th July 2021 Shift 1 - Chemistry - Question 34
Which one of the following compounds of Group-14 elements is not known?
NTA JEE Main 25th July 2021 Shift 1 - Chemistry - Question 35
Which one among the following resonating structures is not correct?
NTA JEE Main 25th July 2021 Shift 1 - Chemistry - Question 36
An organic compound 'A' C$$_4$$H$$_8$$ on treatment with KMnO$$_4$$/H$$^+$$ yields compound 'B' C$$_3$$H$$_6$$O. Compound 'A' also yields compound 'B' on ozonolysis. Compound 'A' is:
NTA JEE Main 25th July 2021 Shift 1 - Chemistry - Question 37
Which one of the following chemical agent is not being used for dry-cleaning of clothes?
NTA JEE Main 25th July 2021 Shift 1 - Chemistry - Question 38
For the following graphs,
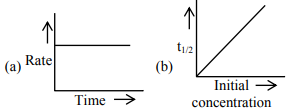
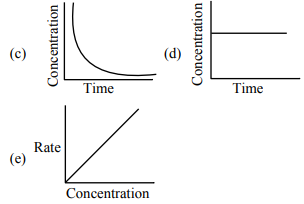
Choose from the options given below, the correct one regarding order of reaction is:
NTA JEE Main 25th July 2021 Shift 1 - Chemistry - Question 39
In the leaching of alumina from bauxite, the ore expected to leach out in the process by reacting with NaOH is:
NTA JEE Main 25th July 2021 Shift 1 - Chemistry - Question 40
The correct order of following 3d metal oxides, according to their oxidation numbers is:
(a) CrO$$_3$$ (b) Fe$$_2$$O$$_3$$ (c) MnO$$_2$$ (d) V$$_2$$O$$_5$$ (e) Cu$$_2$$O
NTA JEE Main 25th July 2021 Shift 1 - Chemistry - Question 41
Which one of the following species responds to an external magnetic field?
NTA JEE Main 25th July 2021 Shift 1 - Chemistry - Question 42
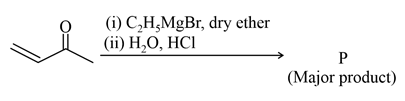
Consider the above reaction, the major product 'P' is:
NTA JEE Main 25th July 2021 Shift 1 - Chemistry - Question 43
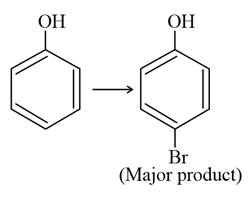
The given reaction can occur in the presence of:
(a) Bromine water
(b) $$Br_{2}$$ in $$CS_{2}$$, 273 K
(c) $$Br_{2} / FeBr_{3}$$
(d) $$Br_{2}$$ in $$CHCl_{3}$$, 273 K
Choose the correct answer from the options given below:
NTA JEE Main 25th July 2021 Shift 1 - Chemistry - Question 44
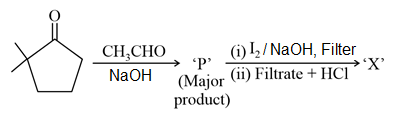
Consider the given reaction, the product 'X' is:
NTA JEE Main 25th July 2021 Shift 1 - Chemistry - Question 45
Given below are two statements, one is labelled as Assertion (A) and other is labelled as Reason (R).
Assertion (R) : Gabriel phthalimide synthesis cannot be used to prepare aromatic primary amines.
Reason : Aryl halides do not undergo nucleophilic substitution reaction.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Main 25th July 2021 Shift 1 - Chemistry - Question 46
Which one of the products of the following reactions does not react with Hinsberg reagent to form sulphonamide?
NTA JEE Main 25th July 2021 Shift 1 - Chemistry - Question 47
Which one of the following compounds will liberate CO$$_2$$, when treated with NaHCO$$_3$$?
NTA JEE Main 25th July 2021 Shift 1 - Chemistry - Question 48
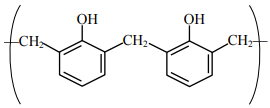
is a repeating unit for:
NTA JEE Main 25th July 2021 Shift 1 - Chemistry - Question 49
Sodium stearate CH$$_3$$CH$$_{2_{16}}$$COO$$^-$$Na$$^+$$ is an anionic surfactant which forms micelles in oil. Choose the correct statement for it from the following:
NTA JEE Main 25th July 2021 Shift 1 - Chemistry - Question 50
The water soluble protein is:
NTA JEE Main 25th July 2021 Shift 1 - Chemistry - Question 51
Consider the complete combustion of butane, the amount of butane utilized to produce 72.0 g of water is ___ $$\times 10^{-1}$$ g. (in nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2021 Shift 1 - Chemistry - Question 52
A source of monochromatic radiation wavelength 400 nm provides 1000 J of energy in 10 seconds. When this radiation falls on the surface of sodium, $$x \times 10^{20}$$ electrons are ejected per second. Assume that wavelength 400 nm is sufficient for ejection of electron from the surface of sodium metal. The value of x is ___. (Nearest integer)
h = 6.626 $$\times 10^{-34}$$ Js
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2021 Shift 1 - Chemistry - Question 53
A home owner uses $$4.00 \times 10^3$$ m$$^3$$ of methane CH$$_4$$ gas, (assume CH$$_4$$ is an ideal gas) in a year to heat his home. Under the pressure of 1.0 atm and 300 K, mass of gas used is $$x \times 10^5$$ g. The value of x is ___.
(Nearest integer)
(Given R = 0.083 L atm K$$^{-1}$$ mol$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2021 Shift 1 - Chemistry - Question 54
At 298 K, the enthalpy of fusion of a solid X is 2.8 kJ mol$$^{-1}$$ and the enthalpy of vaporisation of the liquid X is 98.2 kJ mol$$^{-1}$$. The enthalpy of sublimation of the substance X in kJ mol$$^{-1}$$ is ___. (in nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2021 Shift 1 - Chemistry - Question 55
For the reaction
A + B $$\rightleftharpoons$$ 2C
the value of equilibrium constant is 100 at 298 K. If the initial concentration of all the three species is 1 M each, then the equilibrium concentration of C is $$x \times 10^{-1}$$ M. The value of x is ___. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2021 Shift 1 - Chemistry - Question 56
When 10 mL of an aqueous solution of Fe$$^{2+}$$ ions was titrated in the presence of dil H$$_2$$SO$$_4$$ using diphenylamine indicator, 15 mL of 0.02 M solution of K$$_2$$Cr$$_2$$O$$_7$$ was required to get the end point. The molarity of the solution containing Fe$$^{2+}$$ ions is $$x \times 10^{-2}$$ M. The value of x is ___. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2021 Shift 1 - Chemistry - Question 57
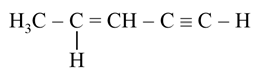
The number of sigma bonds in the above molecule is ___.
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2021 Shift 1 - Chemistry - Question 58
CO$$_2$$ gas is bubbled through water during a soft drink manufacturing process at 298 K. If CO$$_2$$ exerts a partial pressure of 0.835 bar then x m mol of CO$$_2$$ would dissolve in 0.9 L of water. The value of x is ___.
(Nearest integer)
(Henry's law constant for CO$$_2$$ at 298 K is $$1.67 \times 10^3$$ bar)
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2021 Shift 1 - Chemistry - Question 59
Consider the cell at 25$$^\circ$$C
Zn|Zn$$^{2+}$$ aq, 1 M || Fe$$^{3+}$$(aq), Fe$$^{2+}$$aq|Pt
The fraction of total iron present as Fe$$^{3+}$$ ion at the cell potential of 1.500 V is $$x \times 10^{-2}$$. The value of x is ___.
(Nearest integer)
Given: E$$^\circ_{Fe^{3+}|Fe^{2+}}$$ = 0.77V, E$$^\circ_{Zn^{2+}|Zn}$$ = -0.76V
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2021 Shift 1 - Chemistry - Question 60
Three moles of AgCl get precipitated when one mole of an octahedral co-ordination compound with empirical formula CrCl$$_3$$.3NH$$_3$$.3H$$_2$$O reacts with excess of silver nitrate. The number of chloride ions satisfying the secondary valency of the metal ion is ___.
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




