NTA JEE Main 24th June 2022 Shift 1
For the following questions answer them individually
NTA JEE Main 24th June 2022 Shift 1 - Question 41
Given below are the oxides :
$$Na_2O, As_2O_3, N_2O, NO$$ and $$Cl_2O_7$$
Number of amphoteric oxides is :
NTA JEE Main 24th June 2022 Shift 1 - Question 42
The most stable trihalide of nitrogen is :
NTA JEE Main 24th June 2022 Shift 1 - Question 43
Which one of the following elemental forms is not present in the enamel of the teeth?
NTA JEE Main 24th June 2022 Shift 1 - Question 44
Match List - I with List - II :
| List-I | List-II | ||
|---|---|---|---|
| (A) | $$[PtCl_4]^{2-}$$ | (I) | $$sp^3d$$ |
| (B) | $$BrF_5$$ | (II) | $$d^2sp^3$$ |
| (C) | $$PCl_5$$ | (III) | $$dsp^2$$ |
| (D) | $$[Co(NH_3)_6]^{3+}$$ | (IV) | $$sp^3d^2$$ |
Choose the most appropriate answer from the options given below
NTA JEE Main 24th June 2022 Shift 1 - Question 45
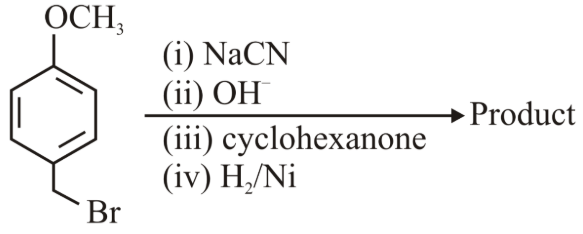
The major product of the above reactions is
NTA JEE Main 24th June 2022 Shift 1 - Question 46
Two statements are given below :
Statement I : The melting point of monocarboxylic acid with even number of carbon atoms is higher than that of with odd number of carbon atoms acid immediately below and above it in the series.
Statement II : The solubility of monocarboxylic acids in water decreases with increase in molar mass.
Choose the most appropriate option :
NTA JEE Main 24th June 2022 Shift 1 - Question 47
Which of the following is an example of polyester?
NTA JEE Main 24th June 2022 Shift 1 - Question 48
Which of the following is not a broad spectrum antibiotic?
NTA JEE Main 24th June 2022 Shift 1 - Question 49
During the qualitative analysis of salt with cation $$y^{2+}$$, addition of a reagent (X) to alkaline solution of the salt gives a bright red precipitate. The reagent (X) and the cation ($$y^{2+}$$) present respectively are :
NTA JEE Main 24th June 2022 Shift 1 - Question 50
A polysaccharide 'X' on boiling with dil $$H_2SO_4$$ at $$393$$ K under $$2 - 3$$ atm pressure yields 'Y' 'Y' on treatment with bromine water gives gluconic acid. 'X' contains $$\beta$$-glycosidic linkages only. Compound 'X' is :


