NTA JEE Main 24th June 2022 Shift 1
For the following questions answer them individually
NTA JEE Main 24th June 2022 Shift 1 - Question 51
$$2O_3(g) \rightleftharpoons 3O_2(g)$$
At $$300$$ K, ozone is fifty percent dissociated. The standard free energy change at this temperature and $$1$$ atm pressure is $$(-)$$ ______ J mol$$^{-1}$$. (Nearest integer)
[Given: $$\ln 1.35 = 0.3$$ and $$R = 8.3$$ J K$$^{-1}$$ mol$$^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Main 24th June 2022 Shift 1 - Question 52
A $$0.166$$ g sample of an organic compound was digested with conc. $$H_2SO_4$$ and then distilled with NaOH. The ammonia gas evolved was passed through $$50.0$$ mL of $$0.5$$ N $$H_2SO_4$$. The used acid required $$30.0$$ mL of $$0.25$$ N NaOH for complete neutralization. The mass percentage of nitrogen in the organic compound is ______
789
456
123
0.-
Clear All
NTA JEE Main 24th June 2022 Shift 1 - Question 53
Number of electrophilic centres in the given compound is ______
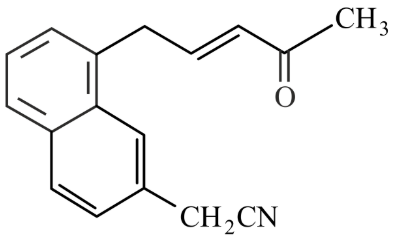
789
456
123
0.-
Clear All
NTA JEE Main 24th June 2022 Shift 1 - Question 54
The major product 'A' of the following given reaction has $$sp^2$$ hybridized carbon atoms.
$$2,7\text{-Dimethyl-2,6-octadiene} \xrightarrow{H^+} A$$ (Major Product)
The number of $$sp^2$$ hybridized carbon atoms in 'A' is ______
789
456
123
0.-
Clear All
NTA JEE Main 24th June 2022 Shift 1 - Question 55
Atoms of element X form hcp lattice and those of element Y occupy $$\frac{2}{3}$$ of its tetrahedral voids. The percentage of element X in the lattice is (Nearest integer) ______
789
456
123
0.-
Clear All
NTA JEE Main 24th June 2022 Shift 1 - Question 56
The osmotic pressure of blood is $$7.47$$ bar at $$300$$ K. To inject glucose to a patient intravenously, it has to be isotonic with blood. The concentration of glucose solution in gL$$^{-1}$$ is (Molar mass of glucose $$= 180$$ g mol$$^{-1}$$, $$R = 0.083$$ Lbar$$^{-1}$$ mol$$^{-1}$$) (Nearest integer) ______
789
456
123
0.-
Clear All
NTA JEE Main 24th June 2022 Shift 1 - Question 57
The cell potential for the following cell $$Pt|H_2(g)|H^+(aq)||Cu^{2+}(0.01 \text{ M})|Cu(s)$$ is $$0.576$$ V at $$298$$ K. The pH of the solution is (Nearest integer) ______
(Given : $$E^{\circ}_{Cu^{2+}/Cu} = 0.34$$ V and $$\frac{2.303RT}{F} = 0.06$$ V)
789
456
123
0.-
Clear All
NTA JEE Main 24th June 2022 Shift 1 - Question 58
The rate constants for decomposition of acetaldehyde have been measured over the temperature range $$700 - 1000$$ K. The data has been analysed by plotting $$\ln k$$ vs $$\frac{10^3}{T}$$ graph. The value of activation energy for the reaction is ______ kJ mol$$^{-1}$$. (Nearest integer) (Given : $$R = 8.31$$ J K$$^{-1}$$ mol$$^{-1}$$)
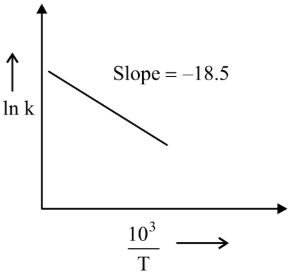
789
456
123
0.-
Clear All
NTA JEE Main 24th June 2022 Shift 1 - Question 59
The difference in oxidation state of chromium in chromate and dichromate salts is ______
789
456
123
0.-
Clear All
NTA JEE Main 24th June 2022 Shift 1 - Question 60
In the cobalt-carbonyl complex : $$[Co_2(CO)_8]$$, number of Co $$-$$ Co bonds is "X" and terminal CO ligands is "Y". $$X + Y =$$ ______
789
456
123
0.-
Clear All


