NTA JEE Main 24th June 2022 Shift 1
For the following questions answer them individually
NTA JEE Main 24th June 2022 Shift 1 - Question 31
If a rocket runs on a fuel ($$C_{15}H_{30}$$) and liquid oxygen, the weight of oxygen required and $$CO_2$$ released for every litre of fuel respectively are :
(Given : density of the fuel is $$0.756$$ g/mL)
NTA JEE Main 24th June 2022 Shift 1 - Question 32
Consider the following pairs of electrons
(A) (a) $$n = 3, l = 1, m_l = 1, m_s = +\frac{1}{2}$$
(b) $$n = 3, l = 2, m_l = 1, m_s = +\frac{1}{2}$$
(B) (a) $$n = 3, l = 2, m_l = -2, m_s = -\frac{1}{2}$$
(b) $$n = 3, l = 2, m_l = -1, m_s = -\frac{1}{2}$$
(C) (a) $$n = 4, l = 2, m_l = 2, m_s = +\frac{1}{2}$$
(b) $$n = 3, l = 2, m_l = 2, m_s = +\frac{1}{2}$$
The pairs of electrons present in degenerate orbitals is/are
NTA JEE Main 24th June 2022 Shift 1 - Question 33
For a reaction at equilibrium
$$A(g) \rightleftharpoons B(g) + \frac{1}{2}C(g)$$
the relation between dissociation constant ($$K$$), degree of dissociation ($$\alpha$$) and equilibrium pressure ($$p$$) is given by :
NTA JEE Main 24th June 2022 Shift 1 - Question 34
The highest industrial consumption of molecular hydrogen is to produce compound of element :
NTA JEE Main 24th June 2022 Shift 1 - Question 35
Which of the following statements are correct?
(A) Both LiCl and MgCl$$_2$$ are soluble in ethanol.
(B) The oxides Li$$_2$$O and MgO combine with excess of oxygen to give superoxide.
(C) LiF is less soluble in water than other alkali metal fluorides.
(D) Li$$_2$$O is more soluble in water than other alkali metal oxides.
Choose the most appropriate answer from the options given below
NTA JEE Main 24th June 2022 Shift 1 - Question 36
Identify the correct statement for $$B_2H_6$$ from those given below.
(A) In $$B_2H_6$$, all B $$-$$ H bonds are equivalent.
(B) In $$B_2H_6$$, there are four 3-centre-2-electron bonds.
(C) $$B_2H_6$$ is a Lewis acid.
(D) $$B_2H_6$$ can be synthesized from both $$BF_3$$ and $$NaBH_4$$.
(E) $$B_2H_6$$ is a planar molecule.
Choose the most appropriate answer from the options given below :
NTA JEE Main 24th June 2022 Shift 1 - Question 37
Which of the following is an example of conjugated diketone?
NTA JEE Main 24th June 2022 Shift 1 - Question 38
In the given reaction sequence, the major product 'C' is :
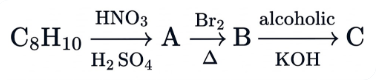
NTA JEE Main 24th June 2022 Shift 1 - Question 39
Given below are two statements:
Statement I : Emulsions of oil in water are unstable and sometimes they separate into two layers on standing.
Statement II : For stabilisation of an emulsion, excess of electrolyte is added.
In the light of the above statements, choose the most appropriate answer from the options given below :
NTA JEE Main 24th June 2022 Shift 1 - Question 40
Match List - I with List - II :
| List-I | List-II | ||
|---|---|---|---|
| (A) | Sphalerite | (I) | FeCO$$_3$$ |
| (B) | Calamine | (II) | PbS |
| (C) | Galena | (III) | ZnCO$$_3$$ |
| (D) | Siderite | (IV) | ZnS |
Choose the most appropriate answer from the options given below :


