NTA JEE Main 24th February 2021 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Main 24th February 2021 Shift 1 - Chemistry - Question 31
Consider the elements Mg, Al, S, P and Si, the correct increasing order of their first ionisation enthalpy is:
NTA JEE Main 24th February 2021 Shift 1 - Chemistry - Question 32
Which of the following are isostructural pairs?
A. $$SO_4^{2-}$$ and $$CrO_4^{2-}$$
B. $$SiCl_4$$ and $$TiCl_4$$
C. $$NH_3$$ and $$NO_3^-$$
D. $$BCl_3$$ and $$BrCl_3$$
NTA JEE Main 24th February 2021 Shift 1 - Chemistry - Question 33
(A) $$HOCl + H_2O_2 \to H_3O^+ + Cl^- + O_2$$
(B) $$I_2 + H_2O_2 + 2OH^- \to 2I^- + 2H_2O + O_2$$
Choose the correct option.
NTA JEE Main 24th February 2021 Shift 1 - Chemistry - Question 34
$$Al_2O_3$$ was leached with alkali to get X. The solution of X on passing of gas Y, forms Z. X, Y and Z respectively are
NTA JEE Main 24th February 2021 Shift 1 - Chemistry - Question 35
Identify products A and B.
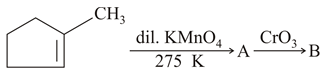
NTA JEE Main 24th February 2021 Shift 1 - Chemistry - Question 36
Which of the following compound gives pink colour on reaction with phthalic anhydride in conc. $$H_2SO_4$$ followed by treatment with NaOH?
NTA JEE Main 24th February 2021 Shift 1 - Chemistry - Question 37
In the following reaction, the reason why meta-nitro product also formed is:
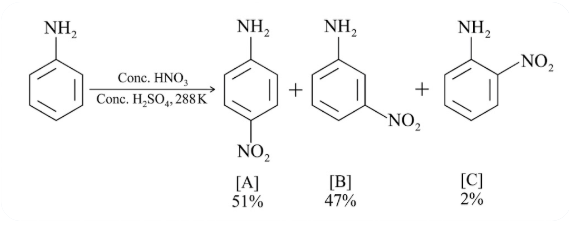
NTA JEE Main 24th February 2021 Shift 1 - Chemistry - Question 38
The gas released during anaerobic degradation of vegetation may lead to:
NTA JEE Main 24th February 2021 Shift 1 - Chemistry - Question 39
In Freundlich adsorption isotherm, slope of AB line is:
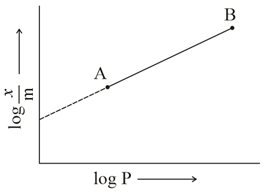
NTA JEE Main 24th February 2021 Shift 1 - Chemistry - Question 40
Which of the following ore is concentrated using group 1 cyanide salt?
NTA JEE Main 24th February 2021 Shift 1 - Chemistry - Question 41
The major components in "Gun Metal" are:
NTA JEE Main 24th February 2021 Shift 1 - Chemistry - Question 42
The electrode potential of $$M^{2+}/M$$ of 3d-series elements shows positive value for?
NTA JEE Main 24th February 2021 Shift 1 - Chemistry - Question 43
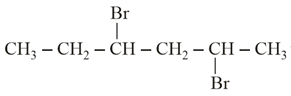
The product formed in the first step of the reaction with excess Mg / Et$$_2$$O / Et = $$C_2H_5$$ is
NTA JEE Main 24th February 2021 Shift 1 - Chemistry - Question 44
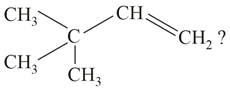
What is the major product formed by HI on reaction with the above compound?
NTA JEE Main 24th February 2021 Shift 1 - Chemistry - Question 45
What is the final product (major) 'A' in the given reaction?
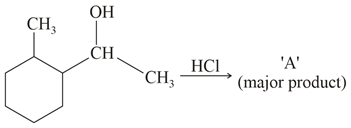
Major product among the following is?
NTA JEE Main 24th February 2021 Shift 1 - Chemistry - Question 46
Which of the following reagent is used for the following reaction?
$$CH_3CH_2CH_3 \to CH_3CH_2CHO$$
NTA JEE Main 24th February 2021 Shift 1 - Chemistry - Question 47
A and B in the following reactions are:
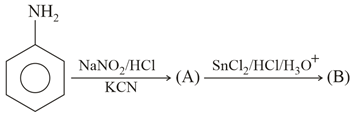
NTA JEE Main 24th February 2021 Shift 1 - Chemistry - Question 48
Match List I with List II.
| List I (Monomer Unit) | List II (Polymer) |
|---|---|
| (a) Caprolactum | (i) Natural rubber |
| (b) 2-Chloro-1,3-butadiene | (ii) Buna-N |
| (c) Isoprene | (iii) Nylon 6 |
| (d) Acrylonitrile | (iv) Neoprene |
Choose the correct answer from the options given below:
NTA JEE Main 24th February 2021 Shift 1 - Chemistry - Question 49
Given below are two statements:
Statement I: Colourless cupric metaborate is reduced to cuprous metaborate in a luminous flame.
Statement II: Cuprous metaborate is obtained by heating boric anhydride and copper sulphate in a non-luminous flame.
In the light of the above statements, choose the most appropriate answer from the options given below.
NTA JEE Main 24th February 2021 Shift 1 - Chemistry - Question 50
Out of the following, which type of interaction is responsible for the stabilisation of $$\alpha$$-helix structure of proteins?
NTA JEE Main 24th February 2021 Shift 1 - Chemistry - Question 51
4.5 g of compound A (M.W. = 90) was used to make 250 mL of its aqueous solution. The molarity of the solution in M is $$x \times 10^{-1}$$. The value of $$x$$ is ______ (Rounded off to the nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 24th February 2021 Shift 1 - Chemistry - Question 52
A proton and a $$Li^{3+}$$ nucleus are accelerated by the same potential. If $$\lambda_{Li}$$ and $$\lambda_p$$ denote the de Broglie wavelengths of $$Li^{3+}$$ and proton respectively, then the value of $$\frac{\lambda_{Li}}{\lambda_p}$$ is $$x \times 10^{-1}$$. The value of $$x$$ is ______ (Rounded off to the nearest integer) [Mass of $$Li^{3+}$$ = 8.3 mass of proton]
789
456
123
0.-
Clear All
NTA JEE Main 24th February 2021 Shift 1 - Chemistry - Question 53
For the reaction $$A_g \to B_g$$, the value of the equilibrium constant at 300 K and 1 atm is equal to 100.0. The value of $$\Delta G^o$$ for the reaction at 300 K and 1 atm in J mol$$^{-1}$$ is $$-xR$$, where $$x$$ is ______ (Rounded off to the nearest integer) R = 8.31 J mol$$^{-1}$$ K$$^{-1}$$ and ln10 = 2.3
789
456
123
0.-
Clear All
NTA JEE Main 24th February 2021 Shift 1 - Chemistry - Question 54
The stepwise formation of $$[Cu(NH_3)_4]^{2+}$$ is given below:
$$Cu^{2+} + NH_3 \xrightleftharpoons{K_1} [Cu(NH_3)]^{2+}$$
$$[Cu(NH_3)]^{2+} + NH_3 \xrightleftharpoons{K_2} [Cu(NH_3)_2]^{2+}$$
$$[Cu(NH_3)_2]^{2+} + NH_3 \xrightleftharpoons{K_3} [Cu(NH_3)_3]^{2+}$$
$$[Cu(NH_3)_3]^{2+} + NH_3 \xrightleftharpoons{K_4} [Cu(NH_3)_4]^{2+}$$
The value of stability constants $$K_1$$, $$K_2$$, $$K_3$$ and $$K_4$$ are $$10^4$$, $$1.58 \times 10^3$$, $$5 \times 10^2$$ and $$10^2$$ respectively. The overall equilibrium constant for dissociation of $$[Cu(NH_3)_4]^{2+}$$ is $$x \times 10^{-12}$$. The value of $$x$$ is ______ (Rounded off to the nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 24th February 2021 Shift 1 - Chemistry - Question 55
At 1990 K and 1 atm pressure, there are equal number of $$Cl_2$$ molecules and Cl atoms in the reaction mixture. The value of $$K_p$$ for the reaction $$Cl_{2g} = 2Cl_g$$ under the above conditions is $$x \times 10^{-1}$$. The value of $$x$$ is ______ (Rounded off to the nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 24th February 2021 Shift 1 - Chemistry - Question 56
The reaction of sulphur in alkaline medium is given below:
$$S_{8s} + a \; OH^-_{aq} \to b \; S^{2-}_{aq} + c \; S_2O^{2-}_{3aq} + d \; H_2O_l$$
The values of 'a' is ______ (Integer answer)
789
456
123
0.-
Clear All
NTA JEE Main 24th February 2021 Shift 1 - Chemistry - Question 57
Number of amphoteric compounds among the following is ______
(A) BeO
(B) BaO
(C) $$BeOH_2$$
(D) $$Sr(OH)_2$$
789
456
123
0.-
Clear All
NTA JEE Main 24th February 2021 Shift 1 - Chemistry - Question 58
The coordination number of an atom in a body-centered cubic structure is ______
[Assume that the lattice is made up of atoms.]
789
456
123
0.-
Clear All
NTA JEE Main 24th February 2021 Shift 1 - Chemistry - Question 59
When 9.45 g of $$ClCH_2COOH$$ is added to 500 mL of water, its freezing point drops by 0.5°C. The dissociation constant of $$ClCH_2COOH$$ is $$x \times 10^{-3}$$. The value of $$x$$ is ______ (off to the nearest integer)
$$K_{f_{H_2O}} = 1.86$$ K kg mol$$^{-1}$$
789
456
123
0.-
Clear All
NTA JEE Main 24th February 2021 Shift 1 - Chemistry - Question 60
Gaseous cyclobutene isomerizes to butadiene in a first order process which has a 'K' value of $$3.3 \times 10^{-4}$$ s$$^{-1}$$ at 153°C. The time in minutes it takes for the isomerization to proceed 40% to completion at this temperature is ______ (Rounded off to the nearest integer)
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




