NTA JEE Main 2025 April 8th Shift 2
For the following questions answer them individually
NTA JEE Main 2025 April 8th Shift 2 - Question 61
The correct decreasing order of spin only magnetic moment values (BM) of $$Cu^+$$, $$Cu^{2+}$$, $$Cr^{2+}$$ and $$Cr^{3+}$$ ions is :
NTA JEE Main 2025 April 8th Shift 2 - Question 62
Which one of the following reactions will not lead to the desired ether formation in major proportion? (iso-Bu $$\Rightarrow$$ isobutyl, sec-Bu $$\Rightarrow$$ sec-butyl, nPr $$\Rightarrow$$ n-propyl, $$^t$$Bu $$\Rightarrow$$ tert-butyl, Et $$\Rightarrow$$ ethyl)
NTA JEE Main 2025 April 8th Shift 2 - Question 63
On combustion 0.210 g of an organic compound containing C, H and O gave 0.127 g $$H_2O$$ and 0.307 g $$CO_2$$. The percentages of hydrogen and oxygen in the given organic compound respectively are :
NTA JEE Main 2025 April 8th Shift 2 - Question 64
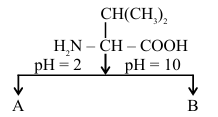
Choose the correct option for structures of A and B, respectively.
NTA JEE Main 2025 April 8th Shift 2 - Question 65
Correct statements for an element with atomic number 9 are :
A. There can be 5 electrons for which $$m_s = +\frac{1}{2}$$ and 4 electrons for which $$m_s = -\frac{1}{2}$$
B. There is only one electron in $$p_z$$ orbital
C. The last electron goes to orbital with $$n = 2$$ and $$l = 1$$
D. The sum of angular nodes of all the atomic orbitals is 1.
Choose the correct answer from the options given below:
NTA JEE Main 2025 April 8th Shift 2 - Question 66
The number of species from the following that are involved in $$sp^3d^2$$ hybridization is : $$[Co(NH_3)_6]^{3+}$$, $$SF_6$$, $$[CrF_6]^{3-}$$, $$[CoF_6]^{3-}$$, $$[Mn(CN)_6]^{3-}$$, and $$[MnCl_6]^{3-}$$
NTA JEE Main 2025 April 8th Shift 2 - Question 67
Match the LIST-I with LIST-II

Choose the correct answer from the options given below :
NTA JEE Main 2025 April 8th Shift 2 - Question 68
When
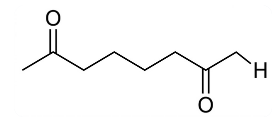
undergoes intramolecular aldol condensation, the major product formed is :
NTA JEE Main 2025 April 8th Shift 2 - Question 69
Match the LIST-I with LIST-II
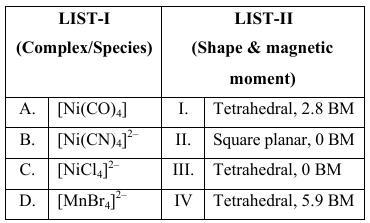
Choose the correct answer from the options given below :
NTA JEE Main 2025 April 8th Shift 2 - Question 70
Given below are two statements :
Statement I : $$H_2Se$$ is more acidic than $$H_2Te$$.
Statement II : $$H_2Se$$ has higher bond enthalpy for dissociation than $$H_2Te$$.
In the light of the above statements, choose the correct answer from the options given below.


