NTA JEE Main 2025 April 8th Shift 2
For the following questions answer them individually
NTA JEE Main 2025 April 8th Shift 2 - Question 71
Resonance in $$X_2Y$$ can be represented as
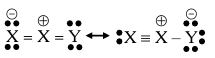
The enthalpy of formation of X_2Y\left( X\equiv X(g)+\frac{1}{2}Y=Y(g)\rightarrow X_2 Y(g)\right) is 80 kJ mol$$^{-1}$$. The magnitude of resonance energy of $$X_2Y$$ is _____ kJ mol$$^{-1}$$ (nearest integer value). Given: Bond energies of $$X \equiv X$$, $$X = X$$, $$Y = Y$$ and $$X = Y$$ are 940, 410, 500 and 602 kJ mol$$^{-1}$$ respectively.
valence X : 3, Y : 2
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 8th Shift 2 - Question 72
The energy of an electron in first Bohr orbit of H-atom is -13.6 eV. The magnitude of energy value of electron in the first excited state of $$Be^{3+}$$ is _____ eV. (nearest integer value)
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 8th Shift 2 - Question 73
20 mL of sodium iodide solution gave 4.74 g silver iodide when treated with excess of silver nitrate solution. The molarity of the sodium iodide solution is _____ M. (Given : Na = 23, I = 127, Ag = 108, N = 14, O = 16 g mol$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 8th Shift 2 - Question 74
The equilibrium constant for decomposition of $$H_2O(g)$$
$$H_2O(g) \rightleftharpoons H_2(g) + \frac{1}{2}O_2(g)$$ ($$\Delta G^\circ = 92.34$$ kJ mol$$^{-1}$$) is $$8.0 \times 10^{-3}$$ at 2300 K and total pressure at equilibrium is 1 bar. Under this condition, the degree of dissociation ($$\alpha$$) of water is _____ $$\times 10^{-2}$$. (nearest integer value)
[Assume $$\alpha$$ is negligible with respect to 1]
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 8th Shift 2 - Question 75
Consider the following half cell reaction: $$Cr_2O_7^{2-}(aq) + 6e^- + 14H^+(aq) \to 2Cr^{3+}(aq) + 7H_2O(l)$$. The reaction was conducted with the ratio of $$\frac{[Cr^{3+}]^2}{[Cr_2O_7^{2-}]} = 10^{-6}$$. The pH value at which the EMF of the half cell will become zero is _____ . (nearest integer value) [Given : standard half cell reduction potential $$E^0_{Cr_{2}O^{2-}_{7},H^{+}/Cr^{3+}} = 1.33$$ V, $$\frac{2.303RT}{F} = 0.059$$ V]
789
456
123
0.-
Clear All


