NTA JEE Main 2025 April 8th Shift 2
For the following questions answer them individually
NTA JEE Main 2025 April 8th Shift 2 - Question 51
In a first order decomposition reaction, the time taken for the decomposition of reactant to one fourth and one eighth of its initial concentration are $$t_1$$ and $$t_2$$ (s), respectively. The ratio $$t_1/t_2$$ will :
NTA JEE Main 2025 April 8th Shift 2 - Question 52
Match the LIST-I with LIST-II.
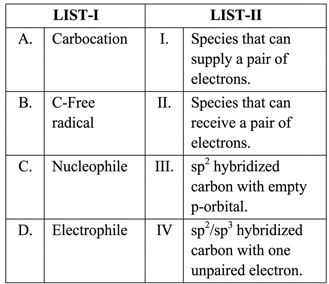
Choose the correct answer from the options given below :
NTA JEE Main 2025 April 8th Shift 2 - Question 53
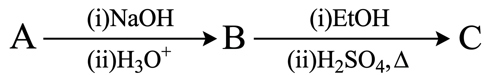
$$A \xrightarrow{(i) NaOH, (ii) H_3O^+} B \xrightarrow{(i) EtOH, (ii) H_2SO_4, \Delta} C$$
'A' shows positive Lassaign's test for N and its molar mass is 121. 'B' gives effervescence with aq. NaHCO$$_3$$. 'C' gives fruity smell. Identify A, B and C.
NTA JEE Main 2025 April 8th Shift 2 - Question 54
Choose the correct set of reagents for the following conversion:
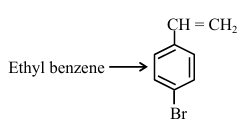
NTA JEE Main 2025 April 8th Shift 2 - Question 55
1,2-dibromocyclooctane is treated with
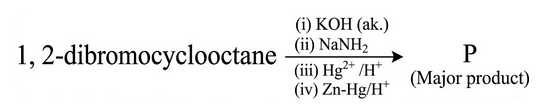
'P' is :
NTA JEE Main 2025 April 8th Shift 2 - Question 56
Given below are two statements :
Statement I : A homoleptic octahedral complex, formed using monodentate ligands, will not show stereoisomerism.
Statement II : cis- and trans-platin are heteroleptic complexes of Pd.
Choose the correct answer from the options given below.
NTA JEE Main 2025 April 8th Shift 2 - Question 57
The atomic number of the element from the following with lowest 1st ionisation enthalpy is :
NTA JEE Main 2025 April 8th Shift 2 - Question 58
Which of the following binary mixture does not show the behaviour of minimum boiling azeotropes?
NTA JEE Main 2025 April 8th Shift 2 - Question 59
$$HA(aq) \rightleftharpoons H^+(aq) + A^-(aq)$$. The freezing point depression of a 0.1 m aqueous solution of a monobasic weak acid HA is 0.20°C. The dissociation constant for the acid is : (Given: $$K_f(H_2O) = 1.8$$ K kg mol$$^{-1}$$, molality $$\equiv$$ molarity)
NTA JEE Main 2025 April 8th Shift 2 - Question 60
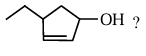
What is the correct IUPAC name of