NTA JEE Main 2025 April 7th Shift 2
For the following questions answer them individually
NTA JEE Main 2025 April 7th Shift 2 - Question 61
The descending order of basicity of following amines is :
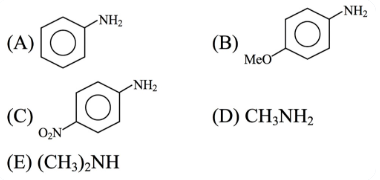
Choose the correct answer from the options given below :
NTA JEE Main 2025 April 7th Shift 2 - Question 62
Match List-I with List-II.
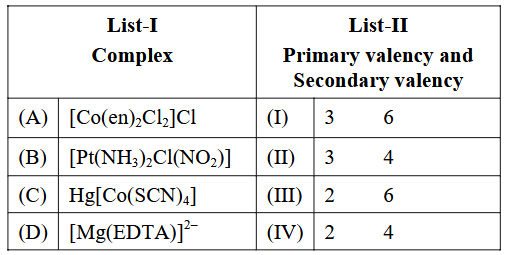
Choose the correct answer from the options given below :
NTA JEE Main 2025 April 7th Shift 2 - Question 63
Match List-I with List-II.
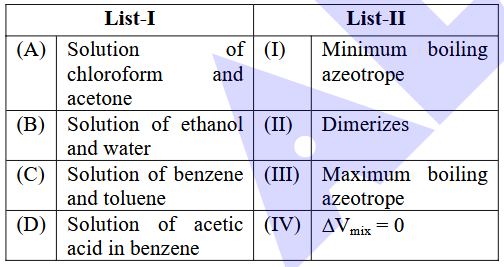
Choose the correct answer from the options given below :
NTA JEE Main 2025 April 7th Shift 2 - Question 64
In $$SO_2$$, $$NO_2^-$$ and $$N_3^-$$ the hybridizations at the central atom are respectively :
NTA JEE Main 2025 April 7th Shift 2 - Question 65
The number of unpaired electrons responsible for the paramagnetic nature of the following species are respectively : $$[Fe(CN)_6]^{3-}$$, $$[FeF_6]^{3-}$$, $$[CoF_6]^{3-}$$, $$[Mn(CN)_6]^{3-}$$
NTA JEE Main 2025 April 7th Shift 2 - Question 66
The number of optically active products obtained from the complete ozonolysis of the given compound is :
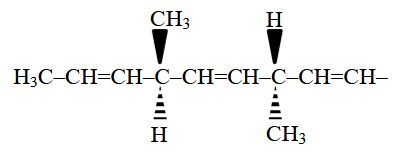
NTA JEE Main 2025 April 7th Shift 2 - Question 67
Given below are two statements :
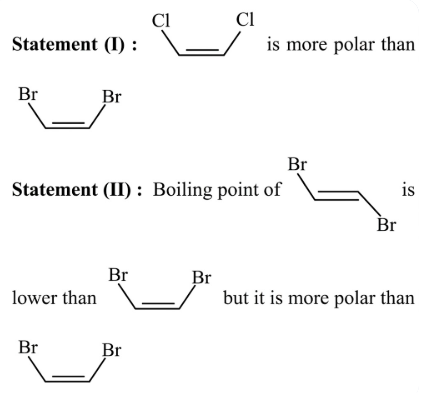
In the light of the above statements, choose the most appropriate answer from the options given below :
NTA JEE Main 2025 April 7th Shift 2 - Question 68
The extra stability of half-filled subshell is due to :
(A) Symmetrical distribution of electrons
(B) Smaller coulombic repulsion energy
(C) The presence of electrons with the same spin in non-degenerate orbitals
(D) Larger exchange energy
(E) Relatively smaller shielding of electrons by one another
Identify the correct statements
NTA JEE Main 2025 April 7th Shift 2 - Question 69
The correct statements from the following are :
(A) $$Tl^{3+}$$ is a powerful oxidising agent
(B) $$Al^{3+}$$ does not get reduced easily
(C) Both $$Al^{3+}$$ and $$Tl^{3+}$$ are very stable in solution
(D) $$Tl^+$$ is more stable than $$Tl^{3+}$$
(E) $$Al^{3+}$$ and $$Tl^+$$ are highly stable
NTA JEE Main 2025 April 7th Shift 2 - Question 70
Given below are two statements :
1 M aqueous solution of each of $$Cu(NO_3)_2$$, $$AgNO_3$$, $$Hg_2(NO_3)_2$$; $$Mg(NO_3)_2$$ are electrolysed using inert electrodes.
Given : $$E_{Ag^{+}/Ag}^{\theta} = 0.80V, E_{Hg_{2}^{2+}/Hg}^{\theta} = 0.79V,$$
$$E_{Cu^{2+}/Cu}^{\theta} = 0.24V$$ and $$E_{Mg^{2+}/Mg}^{\theta} = -2.37V$$
Statement (I) : With increasing voltage, the sequence of deposition of metals on the cathode will be Ag, Hg and Cu
Statement (II) : Magnesium will not be deposited at cathode instead oxygen gas will be evolved at the cathode.
In the light of the above statement, choose the most appropriate answer from the options given below :


