NTA JEE Main 2025 April 7th Shift 2
For the following questions answer them individually
NTA JEE Main 2025 April 7th Shift 2 - Question 71
Only litre buffer solution was prepared by adding 0.10 mol each of $$NH_3$$ and $$NH_4Cl$$ in deionised water. The change in pH on addition of 0.05 mol of HCl to the above solution is _____ $$\times 10^{-2}$$.(Nearest integer)
(Given : $$pK_b$$ of $$NH_3$$ = 4.745 and $$\log_{10}3 = 0.477$$)
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 7th Shift 2 - Question 72
In Dumas' method 292 mg of an organic compound released 50 mL of nitrogen gas ($$N_2$$) at 300 K temperature and 715 mm Hg pressure. The percentage composition of 'N' in the organic compound is _____ %.(Nearest integer)
(Aqueous tension at 300 K = 15 mm Hg)
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 7th Shift 2 - Question 73
Butane reacts with oxygen to produce carbon dioxide and water following the equation given below
$$C_4H_{10}(g) + \frac{13}{2}O_2(g) \to 4CO_2(g) + 5H_2O(l)$$.
If 174.0 kg of butane is mixed with 320.0 kg of $$O_2$$, the volume of water formed in litres is _____.(Nearest integer)
[Given : (a) Molar mass of C, H, O are 12, 1, 16 g $$mol^{-1}$$ respectively, (b) Density of water = 1 g $$mL^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 7th Shift 2 - Question 74
The number of paramagnetic metal complex species among $$[Co(NH_3)_6]^{3+}$$, $$[Co(C_2O_4)_3]^{3-}$$, $$[MnCl_6]^{3-}$$, $$[Mn(CN)_6]^{3-}$$, $$[CoF_6]^{3-}$$, $$[Fe(CN)_6]^{3-}$$ and $$[FeF_6]^{3-}$$ with same number of unpaired electrons is _____.
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 7th Shift 2 - Question 75
Identify the structure of the final product (D) in the following sequence of reactions:
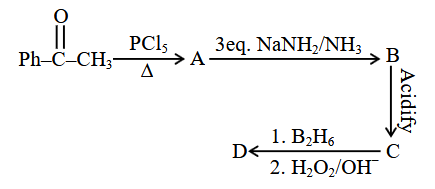
Total number of $$sp^2$$ hybridised carbon atoms in product D is _____.
789
456
123
0.-
Clear All


