NTA JEE Main 2025 April 7th Shift 2
For the following questions answer them individually
NTA JEE Main 2025 April 7th Shift 2 - Question 51
Given below are two statements :
Statement (I) : On hydrolysis, oligo peptides give rise to fewer number of $$\alpha$$-amino acids while proteins give rise to a large number of $$\beta$$-amino acids.
Statement (II) : Natural proteins are denatured by acids which convert the water soluble form of fibrous proteins to their water insoluble form.
In the light of the above statements, choose the most appropriate answer from the options given below :
NTA JEE Main 2025 April 7th Shift 2 - Question 52
Mixture of 1 g each of chlorobenzene, aniline and benzoic acid is dissolved in 50 mL ethyl acetate and placed in a separating funnel, 5 M NaOH (30 mL) was added in the same funnel. The funnel was shaken vigorously and then kept aside. The ethyl acetate layer in the funnel contains :
NTA JEE Main 2025 April 7th Shift 2 - Question 53
The hydration energies of $$K^+$$ and $$Cl^-$$ are $$-x$$ and $$-y$$ kJ/mol respectively. If lattice energy of KCl is $$-z$$ kJ/mol, then the heat of solution of KCl is :
NTA JEE Main 2025 April 7th Shift 2 - Question 54
$$A(g) \to B(g) + C(g)$$ is a first order reaction.
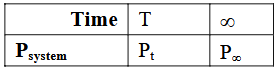
The reaction was started with reactant A only. Which of the following expression is correct for rate constant k?
NTA JEE Main 2025 April 7th Shift 2 - Question 55
"P" is an optically active compound with molecular formula $$C_6H_{12}O$$. When "P" is treated with 2,4-dinitrophenylhydrazine, it gives a positive test. However, in presence of Tollens reagent, "P" gives a negative test. Predict the structure of "P".
NTA JEE Main 2025 April 7th Shift 2 - Question 56
Choose the incorrect trend in the atomic radii (r) of the elements :
NTA JEE Main 2025 April 7th Shift 2 - Question 57
Match List-I with List-II .
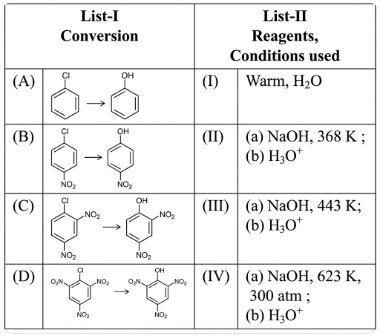
Choose the correct answer from the options given below :
NTA JEE Main 2025 April 7th Shift 2 - Question 58
The correct statement amongst the following is :
NTA JEE Main 2025 April 7th Shift 2 - Question 59
Liquid A and B form an ideal solution. The vapour pressure of pure liquids A and B are 350 and 750 mm Hg respectively at the same temperature. If $$x_A$$ and $$x_B$$ are the mole fraction of A and B in solution while $$y_A$$ and $$y_B$$ are the mole fraction of A and B in vapour phase then :
NTA JEE Main 2025 April 7th Shift 2 - Question 60
'X' is the number of acidic oxides among $$VO_2$$, $$V_2O_3$$, $$CrO_3$$, $$V_2O_5$$ and $$Mn_2O_7$$. The primary valency of cobalt in $$[Co(H_2NCH_2CH_2NH_3)_3]_2(SO_4)_3$$ is Y. The value of X + Y is :


