NTA JEE Main 2025 April 7th Shift 1
For the following questions answer them individually
NTA JEE Main 2025 April 7th Shift 1 - Question 61
The number of valence electrons present in the metal among Cr, Co, Fe and Ni which has the lowest enthalpy of atomisation is
NTA JEE Main 2025 April 7th Shift 1 - Question 62
When a salt is treated with sodium hydroxide solution it gives gas X. On passing gas X through reagent Y a brown coloured precipitate is formed. X and Y respectively, are
NTA JEE Main 2025 April 7th Shift 1 - Question 63
The group 14 elements A and B have the first ionisation enthalpy values of 708 and 715 kJ mol$$^{-1}$$ respectively. The above values are lowest among their group members. The nature of their ions $$A^{2+}$$ $$B^{4+}$$ respectively is
NTA JEE Main 2025 April 7th Shift 1 - Question 64
The first transition series metal 'M' has the highest enthalpy of atomisation in its series. One of its aquated ion ($$M^{n+}$$) exists in green colour. The nature of the oxide formed by the above $$M^{n-}$$ ion is :
NTA JEE Main 2025 April 7th Shift 1 - Question 65
Which of the following compounds is least likely to give effervescence of $$CO_2$$ in presence of aq. NaHCO$$_3$$?
NTA JEE Main 2025 April 7th Shift 1 - Question 66
Match the LIST-I with LIST-II.
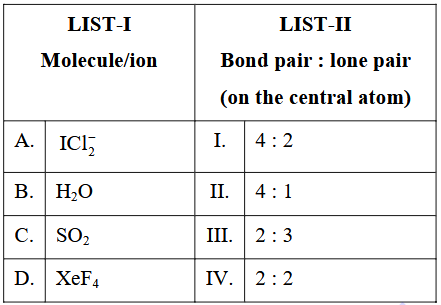
Choose the correct answer from the options given below :
NTA JEE Main 2025 April 7th Shift 1 - Question 67
A person's wound was exposed to some bacteria and then bacteria growth started to happen at the same place. The wound was later treated with some antibacterial medicine and the rate of bacterial decay (r) was found to be proportional with the square of the existing number of bacteria at any instance. Which of the following set of graphs correctly represents the 'before' and 'after' situation of the application of the medicine?
[Given : N = No. of bacteria, t = time, bacteria growth follows $$I^{st}$$ order kinetics.]
NTA JEE Main 2025 April 7th Shift 1 - Question 68
Given below are two statements :
Statement I: D-(+)-glucose + D-(+)-fructose $$\xrightarrow{-H_2O}$$ sucrose
sucrose $$\xrightarrow{Hydrolysis}$$ D-(+)-glucose + D-(+)-fructose
Statement II : Invert sugar is formed during sucrose hydrolysis.
In the light of the above statements, choose the correct answer from the options given below-
NTA JEE Main 2025 April 7th Shift 1 - Question 69
An octahedral complex having molecular composition $$Co \cdot 5NH_3 \cdot Cl \cdot SO_4$$ has two isomers A and B. The solution of A gives a white precipitate with $$AgNO_3$$ solution and the solution of B gives white precipitate with $$BaCl_2$$ solution. The type of isomerism exhibited by the complex is,
NTA JEE Main 2025 April 7th Shift 1 - Question 70
The reactions which cannot be applied to prepare an alkene by elimination, are
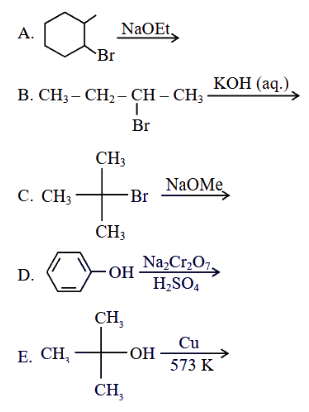
Choose the correct answer from the options given below :


