NTA JEE Main 2025 April 7th Shift 1
For the following questions answer them individually
NTA JEE Main 2025 April 7th Shift 1 - Question 51
Given below are two statements :
Statement I : Ozonolysis followed by treatment with Zn, H$$_2$$O of cis-2-butene gives ethanal.
Statement II : The production obtained by ozonolysis followed by treatment with Zn, H$$_2$$O of 3, 6-dimethyloct-4-ene has no chiral carbon atom.
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Main 2025 April 7th Shift 1 - Question 52
Which of the following amine(s) show(s) positive carbylamines test?
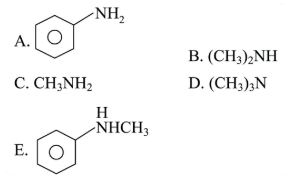
Choose the correct answer from the options given below :
NTA JEE Main 2025 April 7th Shift 1 - Question 53
Reaction $$A(g) \rightarrow 2B(g) + C(g)$$ is a first order reaction. It was started with pure A.

Which of the following option is incorrect?
NTA JEE Main 2025 April 7th Shift 1 - Question 54
Total enthalpy change for freezing of 1 mol water at 10 degrees C to ice at -10 degrees C is ______
(Given : $$\Delta_{fus}H = x$$ kJ/mol, $$C_p[H_2O(l)] = y$$ J mol$$^{-1}$$ K$$^{-1}$$, $$C_p[H_2O(s)] = z$$ J mol$$^{-1}$$ K$$^{-1}$$)
NTA JEE Main 2025 April 7th Shift 1 - Question 55
An aqueous solution of HCl with pH 1.0 is diluted by adding equal volume of water (ignoring dissociation of water). The pH of HCl solution would
(Given log 2 = 0.30)
NTA JEE Main 2025 April 7th Shift 1 - Question 56
Given below are two statements :
Statement I : Dimethyl ether is completely soluble in water. However, diethyl ether is soluble in water to a very small extent.
Statement II : Sodium metal can be used to dry diethyl ether and not ethyl alcohol.
In the light of given statements, choose the correct answer from the options given below
NTA JEE Main 2025 April 7th Shift 1 - Question 57
Which of the following statements are correct, if the threshold frequency of caesium is $$5.16 \times 10^{14}$$ Hz?
A. When Cs is placed inside a vacuum chamber with an ammeter connected to it and yellow light is focused on Cs the ammeter shows the presence of current.
B. When the brightness of the yellow light is dimmed, the value of the current in the ammeter is reduced.
C. When a red light is used instead to the yellow light, the current produced is higher with respect to the yellow light.
D. When a blue light is used, the ammeter shows the formation of current.
E. When a white light is used, the ammeter shows formation of current.
NTA JEE Main 2025 April 7th Shift 1 - Question 58
Which of the following is the correct IUPAC name of given organic compound (X)?
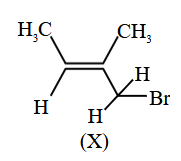
NTA JEE Main 2025 April 7th Shift 1 - Question 59
At the sea level, the dry air mass percentage composition is given as nitrogen gas : 70.0, oxygen gas : 27.0 and argon gas : 3.0. If total pressure is 1.15 atm, then calculate the ratio of followings respectively :
(i) partial pressure of nitrogen gas to partial pressure of oxygen gas
(ii) partial pressure of oxygen gas to partial pressure of argon gas
(Given : Molar mass of $$N, O$$ and $$Ar$$ are 14, 16, and 40 g $$mol^{-1}$$ respectively)
NTA JEE Main 2025 April 7th Shift 1 - Question 60
Given below are two statements :
Statement I : Mohr's salt is composed of only three types of ions-ferrous, ammonium and sulphate.
Statement II : If the molar conductance at infinite dilution of ferrous, ammonium and sulphate ions are $$x_1$$, $$x_2$$ and $$x_3$$ S cm$$^2$$ mol$$^{-1}$$, respectively then the molar conductance for Mohr's salt solution at infinite dilution would be given by $$x_1 + x_2 + 2x_3$$
In the light of the given statements, choose the correct answer from the options given below :


