NTA JEE Main 2025 April 7th Shift 1
For the following questions answer them individually
NTA JEE Main 2025 April 7th Shift 1 - Question 71
An organic compound weighing 500 mg, produced 220 mg of $$CO_2$$ on complete combustion. The percentage composition of carbon in the compound is .......... %. (nearest integer)
(Given molar mass in g $$mol^{-1}$$ of C : 12, O : 16)
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 7th Shift 1 - Question 72
Thyroxine, the hormone has the given below structure.
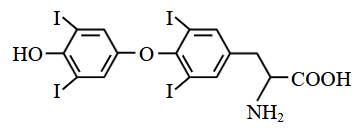
The percentage of iodine in thyroxine is .......... %. (nearest integer)
(Given molar mass in g mol$$^{-1}$$ of C:12, H:1, O:16, N:14, I:127)
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 7th Shift 1 - Question 73
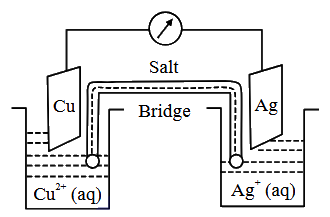
1 Faraday electricity was passed through $$Cu^{2+}$$ (1.5 M, 1 L)/Cu and 0.1 Faraday was passed through $$Ag^+$$ (0.2 M, 1 L)/Ag electrolytic cells. After this the two cells were connected as shown below to make an electrochemical cell. The emf of the cell thus formed at 298 K is ______. (Given: $$E^0_{Cu^{2+}/Cu} = 0.34$$ V, $$E^0_{Ag^+/Ag} = 0.8$$ V, $$\frac{2.303RT}{F} = 0.06$$ V)
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 7th Shift 1 - Question 74
The percentage dissociation of a salt (MX$$_3$$) solution at given temperature (van't Hoff factor i = 2) is .......... % (Nearest Integer)
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 7th Shift 1 - Question 75
The number of paramagnetic complex among $$[FeF_6]^{3-}$$, $$[Fe(CN)_6]^{3-}$$, $$[Mn(CN)_6]^{3-}$$, $$[Co(C_2O_4)_3]^{3-}$$, $$[MnCl_6]^{3-}$$ and $$[CoF_6]^{3-}$$, which involved $$d^2sp^3$$ hybridisation of metal is ______.
789
456
123
0.-
Clear All


