NTA JEE Main 2025 April 02 Shift 2
For the following questions answer them individually
NTA JEE Main 2025 April 02 Shift 2 - Question 61
Match List-I with List-II.
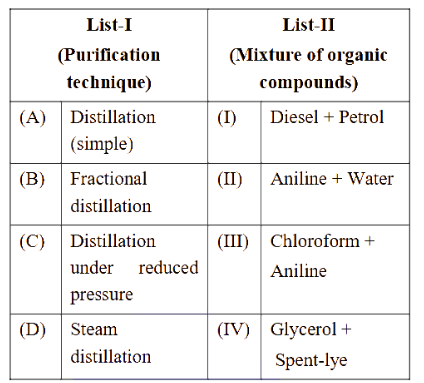
Choose the correct answer from the options given below :
NTA JEE Main 2025 April 02 Shift 2 - Question 62
'x' g of NaCl is added to water in a beaker with a lid. The temperature of the system is raised from 1°C to 25°C. Which of the following plots, is best suited for the change in the molarity (M) of the solution with respect to temperature ?
[Consider the solubility of NaCl remains unchanged over the temperature range]
NTA JEE Main 2025 April 02 Shift 2 - Question 63
Arrange the following in order of magnitude of work done by the system / on the system at constant temperature :
(a) $$|w_{\text{reversible}}|$$ for expansion in infinite stage.
(b) $$|w_{\text{irreversible}}|$$ for expansion in single stage.
(c) $$|w_{\text{reversible}}|$$ for compression in infinite stage.
(d) $$|w_{\text{irreversible}}|$$ for compression in single stage.
Choose the correct answer from the options given below :
NTA JEE Main 2025 April 02 Shift 2 - Question 64
Reactant A converts to product D through the given mechanism (with the net evolution of heat) :
$$A \to B$$ slow ; $$\Delta H = +ve$$
$$B \to C$$ fast ; $$\Delta H = -ve$$
$$C \to D$$ fast ; $$\Delta H = -ve$$
Which of the following represents the above reaction mechanism ?
NTA JEE Main 2025 April 02 Shift 2 - Question 65
The nature of oxide $$(TeO_2)$$ and hydride $$(TeH_2)$$ formed by Te, respectively are :
NTA JEE Main 2025 April 02 Shift 2 - Question 66
Match List-I with List-II.
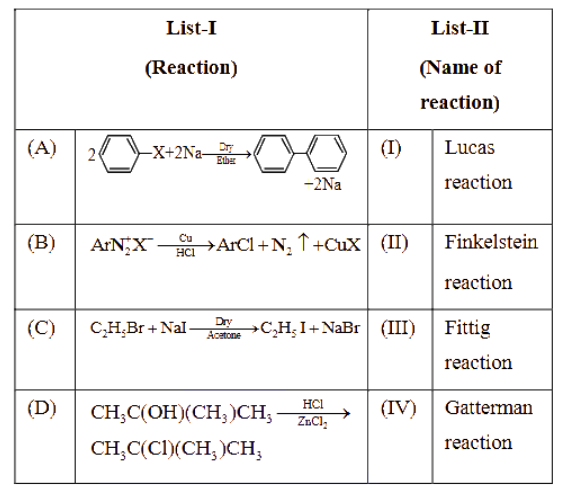
Choose the correct answer from the options given below :
NTA JEE Main 2025 April 02 Shift 2 - Question 67
Consider the following chemical equilibrium of the gas phase reaction at a constant temperature :
$$A(g) \rightleftharpoons B(g) + C(g)$$
If p being the total pressure, $$K_p$$ is the pressure equilibrium constant and $$\alpha$$ is the degree of dissociation, then which of the following is true at equilibrium ?
NTA JEE Main 2025 April 02 Shift 2 - Question 68
Which of the following graphs correctly represents the variation of thermodynamic properties of Haber's process ?
NTA JEE Main 2025 April 02 Shift 2 - Question 69
A tetrapeptide "x" on complete hydrolysis produced glycine (Gly), alanine (Ala), valine (Val), leucine (Leu) in equimolar proportion each. The number of tetrapeptides (sequences) possible involving each of these amino acids is
NTA JEE Main 2025 April 02 Shift 2 - Question 70
In Dumas' method for estimation of nitrogen, 0.5 gram of an organic compound gave 60 mL of nitrogen collected at 300 K temperature and 715 mm Hg pressure. The percentage composition of nitrogen in the compound (Aqueous tension at 300 K = 15 mm Hg) is


