NTA JEE Main 2025 April 02 Shift 2
For the following questions answer them individually
NTA JEE Main 2025 April 02 Shift 2 - Question 51
When a concentrated solution of sulphanilic acid and 1-naphthylamine is treated with nitrous acid (273 K) and acidified with acetic acid, the mass (g) of 0.1 mole of product formed is :
(Given molar mass in g mol$$^{-1}$$ H : 1, C : 12, N : 14, O : 16, S : 32)
NTA JEE Main 2025 April 02 Shift 2 - Question 52
The d-orbital electronic configuration of the complex among $$[Co(en)_3]^{3+}$$, $$[CoF_6]^{3-}$$, $$[Mn(H_2O)_6]^{2+}$$ and $$[Zn(H_2O)_6]^{2+}$$ that has the highest CFSE is :
NTA JEE Main 2025 April 02 Shift 2 - Question 53
Given below are two statements :
Statement (I) : Neopentane forms only one monosubstituted derivative.
Statement (II) : Melting point of neopentane is higher than n-pentane.
In the light of the above statements, choose the most appropriate answer from the options given below :
NTA JEE Main 2025 April 02 Shift 2 - Question 54
Which among the following molecules is (a) involved in $$sp^3d$$ hybridization, (b) has different bond lengths and (c) has lone pair of electrons on the central atom ?
NTA JEE Main 2025 April 02 Shift 2 - Question 55
Formation of $$Na_4[Fe(CN)_5NOS]$$, a purple coloured complex formed by addition of sodium nitroprusside in sodium carbonate extract of salt indicates the presence of :
NTA JEE Main 2025 April 02 Shift 2 - Question 56
In 3,3-dimethylhex-1-ene-4-yne, there are _____ $$sp^3$$, _____ $$sp^2$$ and _____ $$sp$$ hybridised carbon atoms respectively :
NTA JEE Main 2025 April 02 Shift 2 - Question 57
Which of the following statements are true ?
(A) The subsidiary quantum number $$l$$ describes the shape of the orbital occupied by the electron.
(B)
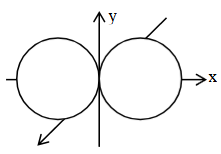
is the boundary diagram of the $$2p_x$$ orbital.
(C) The + and - signs in the wave function of the $$2p_x$$ orbital refer to charge.
(D) The wave function of $$2p_x$$ orbital is zero everywhere in the xy plane.
NTA JEE Main 2025 April 02 Shift 2 - Question 58
The type of hybridization and the magnetic property of $$[MnCl_6]^{3-}$$ are :
NTA JEE Main 2025 April 02 Shift 2 - Question 59
Consider the following reactions. From these reactions which reaction will give carboxylic acid as a major product ?
(A) $$R-C \equiv N \xrightarrow[\text{mild condition}]{(i) H^+/H_2O}$$
(B) $$R-MgX \xrightarrow[(ii) H_3O^+]{(i) CO_2}$$
(C) $$R-C \equiv N \xrightarrow[(ii) H_3O^+]{(i) SnCl_2/HCl}$$
(D) $$R-CH_2-OH \xrightarrow{PCC}$$
(E)
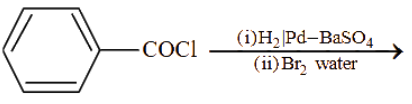
Choose the correct answer from the options given below :
NTA JEE Main 2025 April 02 Shift 2 - Question 60
Electronic configuration of four elements A, B, C and D are given below :
(A) $$1s^2 2s^2 2p^3$$
(B) $$1s^2 2s^2 2p^4$$
(C) $$1s^2 2s^2 2p^5$$
(D) $$1s^2 2s^2 2p^2$$
Which of the following is the correct order of increasing electronegativity (Pauling's scale) ?


