NTA JEE Main 2025 April 02 Shift 2
For the following questions answer them individually
NTA JEE Main 2025 April 02 Shift 2 - Question 71
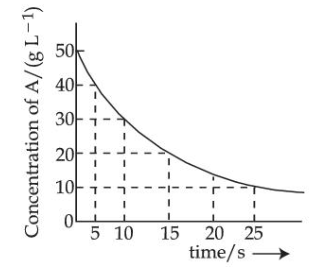
For the reaction $$A \to B$$ the following graph was obtained. The time required (in seconds) for the concentration of A to reduce to 2.5 g L$$^{-1}$$ (if the initial concentration of A was 50 g L$$^{-1}$$) is _________.
(Nearest integer)
Given : $$\log 2 = 0.3010$$
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 02 Shift 2 - Question 72
0.2 % (w/v) solution of NaOH is measured to have resistivity 870.0 m$$\Omega$$ m. The molar conductivity of the solution will be _________ $$\times 10^2$$ mS dm$$^2$$ mol$$^{-1}$$.
(Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 02 Shift 2 - Question 73
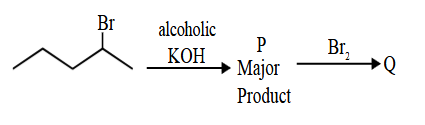
Consider the above sequence of reactions. 151 g of 2-bromopentane is made to react. Yield of major product P is 80% whereas Q is 100%.
Mass of product Q obtained is _________ g.
(Given molar mass in g mol$$^{-1}$$ H: 1, C: 12, O: 16, Br: 80)
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 02 Shift 2 - Question 74
When 1 g each of compounds AB and $$AB_2$$ are dissolved in 15 g of water separately, they increased the boiling point of water by 2.7 K and 1.5 K respectively. The atomic mass of A (in amu) is _________ $$\times 10^{-1}$$.
(Nearest integer)
(Given : Molal boiling point elevation constant is 0.5 K kg mol$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 02 Shift 2 - Question 75
The spin-only magnetic moment value of $$M^{n+}$$ ion formed among Ni, Zn, Mn and Cu that has the least enthalpy of atomisation is _________. (in nearest integer)
Here n is equal to the number of diamagnetic complexes among $$K_2[NiCl_4]$$, $$[Zn(H_2O)_6]Cl_2$$, $$K_3[Mn(CN)_6]$$ and $$[Cu(PPh_3)_3I]$$
789
456
123
0.-
Clear All


