NTA JEE Main 2025 April 02 Shift 1
For the following questions answer them individually
NTA JEE Main 2025 April 02 Shift 1 - Question 61
Given below are two statements:
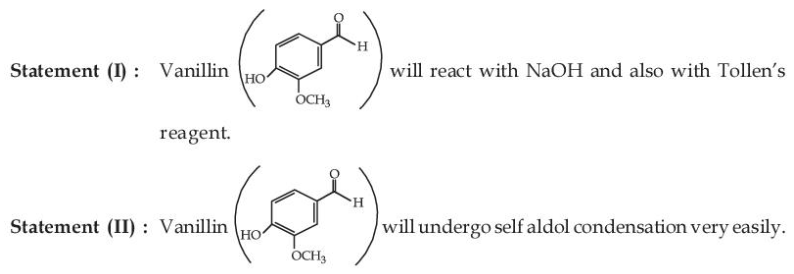
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Main 2025 April 02 Shift 1 - Question 62
Identify the correct statement among the following:
NTA JEE Main 2025 April 02 Shift 1 - Question 63
The correct order of basic nature on aqueous solution for the bases $$NH_3$$, $$H_2N-NH_2$$, $$CH_3CH_2NH_2$$, $$(CH_3CH_2)_2NH$$ and $$(CH_3CH_2)_3N$$ is:
NTA JEE Main 2025 April 02 Shift 1 - Question 64
Given below are two statements:
Statement (I): The metallic radius of Al is less than that of Ga.
Statement (II): The ionic radius of Al$$^{3+}$$ is less than that of Ga$$^{3+}$$.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Main 2025 April 02 Shift 1 - Question 65
Given below are two statements:
Statement (I): In octahedral complexes, when $$\Delta_o \lt P$$ high spin complexes are formed. When $$\Delta_o \gt P$$ low spin complexes are formed.
Statement (II): In tetrahedral complexes because of $$\Delta_t \lt P$$, low spin complexes are rarely formed.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Main 2025 April 02 Shift 1 - Question 66
Choose the correct tests with respective observations.
(A) $$CuSO_4$$ (acidified with acetic acid) + $$K_4[Fe(CN)_6]$$ $$\rightarrow$$ Chocolate brown precipitate.
(B) $$FeCl_3 + K_4[Fe(CN)_6]$$ $$\rightarrow$$ Prussian blue precipitate.
(C) $$ZnCl_2 + K_4[Fe(CN)_6]$$, neutralised with $$NH_4OH$$ $$\rightarrow$$ White or bluish white precipitate.
(D) $$MgCl_2 + K_4[Fe(CN)_6]$$ $$\rightarrow$$ Blue precipitate.
(E) $$BaCl_2 + K_4[Fe(CN)_6]$$, neutralised with NaOH $$\rightarrow$$ White precipitate.
Choose the correct answer from the options given below:
NTA JEE Main 2025 April 02 Shift 1 - Question 67
On complete combustion 1.0 g of an organic compound (X) gave 1.46 g of $$CO_2$$ and 0.567 g of $$H_2O$$. The empirical formula mass of compound (X) is ________ g.
(Given molar mass in g mol$$^{-1}$$ C: 12, H: 1, O: 16)
NTA JEE Main 2025 April 02 Shift 1 - Question 68
Consider the following compound (X):
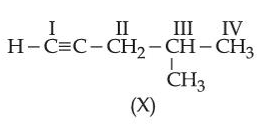
The most stable and least stable carbon radicals, respectively, produced by homolytic cleavage of corresponding C-H bond are:
NTA JEE Main 2025 April 02 Shift 1 - Question 69
Consider the following molecules:
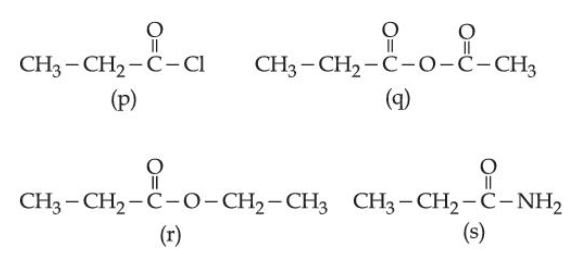
The correct order of rate of hydrolysis is:
NTA JEE Main 2025 April 02 Shift 1 - Question 70
A molecule with the formula $$AX_4Y$$ has all its elements from p-block. Element $$A$$ is rarest, monoatomic, non-radioactive from its group and has the lowest ionization enthalpy value among$$ A, X$$ and $$Y$$. Elements $$X$$ and $$Y$$ have first and second highest electronegativity values respectively among all the known elements. The shape of the molecule is:


