NTA JEE Main 2025 April 02 Shift 1
For the following questions answer them individually
NTA JEE Main 2025 April 02 Shift 1 - Question 71
A transition metal $$(M)$$ among $$Mn, Cr, Co$$ and $$Fe$$ has the highest standard electrode potential ($$M^{3+}/M^{2+}$$). It forms a metal complex of the type $$[M(CN)_6]^{4-}$$. The number of electrons present in the $$e_g$$ orbital of the complex is ______.
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 02 Shift 1 - Question 72
Consider the following electrochemical cell at standard condition.
$$Au(s)|QH_2,Q|NH_4X(0.01M)||Ag^+(1M)|Ag(s)$$
$$E_{cell} = +0.4V$$
The couple $$QH_2/Q$$ represents quinhydrone electrode, the half cell reaction is given below:
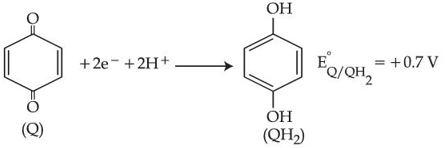
[Given: $$E^o_{Ag^+/Ag} = +0.8V$$ and $$\frac{2.303RT}{F} = 0.06V$$]
The $$pK_b$$ value of the ammonium halide salt ($$NH_4X$$) used here is ______. (nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 02 Shift 1 - Question 73
0.1 mol of the following given antiviral compound (P) will weigh _______ $$\times 10^{-1}$$ g.
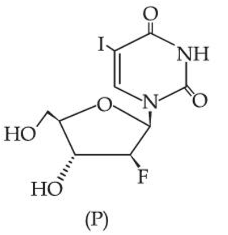
(Given: molar mass in g mol$$^{-1}$$ H: 1, C: 12, N: 14, O: 16, F: 19, I: 127)
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 02 Shift 1 - Question 74
Consider the following equilibrium,
$$CO(g) + 2H_2(g) \rightleftharpoons CH_3OH(g)$$
0.1 mol of CO along with a catalyst is present in a 2 dm$$^3$$ flask maintained at 500 K. Hydrogen is introduced into the flask until the pressure is 5 bar and 0.04 mol of $$CH_3OH$$ is formed. The $$K_p^0$$ is _______ $$\times 10^{-3}$$. (nearest integer)
Given $$R = 0.08$$ dm$$^3$$ bar K$$^{-1}$$ mol$$^{-1}$$
Assume only methanol is formed as the product and the system follows ideal gas behaviour.
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 02 Shift 1 - Question 75
For the reaction $$A \rightarrow$$ products.
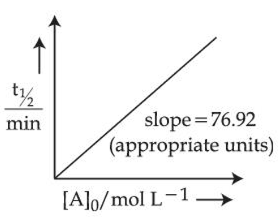
The concentration of A at 10 minutes is _______ $$\times 10^{-3}$$ mol L$$^{-1}$$. (nearest integer).
The reaction was started with 2.5 mol L$$^{-1}$$ of A.
789
456
123
0.-
Clear All


