NTA JEE Main 2025 April 02 Shift 1
For the following questions answer them individually
NTA JEE Main 2025 April 02 Shift 1 - Question 51
Designate whether each of the following compounds is aromatic or not aromatic.
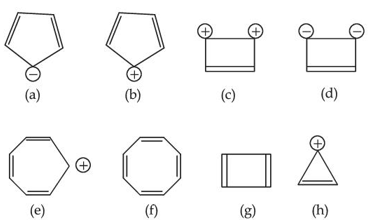
NTA JEE Main 2025 April 02 Shift 1 - Question 52
An optically active alkyl halide $$C_4H_9Br$$ [A] reacts with hot KOH dissolved in ethanol and forms alkene [B] as major product which reacts with bromine to give dibromide [C]. The compound [C] is converted into a gas [D] upon reacting with alcoholic NaNH$$_2$$. During hydration 18 gram of water is added to 1 mole of gas [D] on warming with mercuric sulphate and dilute acid at 333 K to form compound [E]. The IUPAC name of compound [E] is:
NTA JEE Main 2025 April 02 Shift 1 - Question 53
The property/properties that show irregularity in first four elements of group-17 is/are:
(A) Covalent radius
(B) Electron affinity
(C) Ionic radius
(D) First ionization energy
Choose the correct answer from the options given below:
NTA JEE Main 2025 April 02 Shift 1 - Question 54
Which of the following graph correctly represents the plots of $$K_H$$ at 1 bar gases in water versus temperature?
NTA JEE Main 2025 April 02 Shift 1 - Question 55
According to Bohr's model of hydrogen atom, which of the following statement is incorrect?
NTA JEE Main 2025 April 02 Shift 1 - Question 56
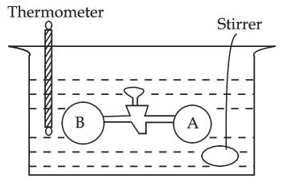
Two vessels A and B are connected via stopcock. The vessel A is filled with a gas at a certain pressure. The entire assembly is immersed in water and is allowed to come to thermal equilibrium with water. After opening the stopcock the gas from vessel A expands into vessel B and no change in temperature is observed in the thermometer. Which of the following statement is true?
NTA JEE Main 2025 April 02 Shift 1 - Question 57
A solution is made by mixing one mole of volatile liquid A with 3 moles of volatile liquid B. The vapour pressure of pure A is 200 mm Hg and that of the solution is 500 mm Hg. The vapour pressure of pure B and the least volatile component of the solution, respectively, are:
NTA JEE Main 2025 April 02 Shift 1 - Question 58
$$CaCO_3(s) + 2HCl(aq) \rightarrow CaCl_2(aq) + CO_2(g) + H_2O(l)$$
Consider the above reaction, what mass of $$CaCl_2$$ will be formed if 250 mL of 0.76 M HCl reacts with 1000 g of $$CaCO_3$$?
(Given: Molar mass of Ca, C, O, H and Cl are 40, 12, 16, 1 and 35.5 g mol$$^{-1}$$, respectively)
NTA JEE Main 2025 April 02 Shift 1 - Question 59
If equal volumes of $$AB_2$$ and XY (both are salts) aqueous solutions are mixed, which of the following combination will give a precipitate of $$AY_2$$ at 300 K?
(Given $$K_{sp}$$ (at 300 K) for $$AY_2 = 5.2 \times 10^{-7}$$)
NTA JEE Main 2025 April 02 Shift 1 - Question 60
Among $$SO_2$$, $$NF_3$$, $$NH_3$$, $$XeF_2$$, $$ClF_3$$ and $$SF_4$$, the hybridization of the molecule with non-zero dipole moment and highest number of lone pairs of electrons on the central atom is:


