JEE Main 12th January 2019 Shift 1
For the following questions answer them individually
JEE Main 12th January 2019 Shift 1 - Question 41
Among the following four aromatic compounds, which one will have the lowest melting point?
JEE Main 12th January 2019 Shift 1 - Question 42
The correct order for acid strength of compounds $$CH \equiv CH$$, $$CH_3 - C \equiv CH$$ and $$CH_2 = CH_2$$ is as follows:
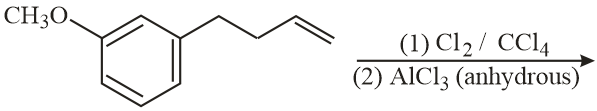
JEE Main 12th January 2019 Shift 1 - Question 43
The major product of the following reaction is:
JEE Main 12th January 2019 Shift 1 - Question 44
Water samples with BOD values of 4 ppm and 18 ppm, respectively, are:
JEE Main 12th January 2019 Shift 1 - Question 45
The molecule that has minimum or no role in the formation of photochemical smog, is:
JEE Main 12th January 2019 Shift 1 - Question 46
The freezing point of a 4% aqueous solution of X is equal to the freezing point of a 12% aqueous solution of Y. If the molecular weight of X is A, then the molecular weight of Y will be
JEE Main 12th January 2019 Shift 1 - Question 47
The standard electrode potential E° and its temperature coefficient $$\left(\frac{dE}{dT}\right)$$ for a cell are 2 V and $$-5 \times 10^{-4}$$ V K$$^{-1}$$ at 300 K, respectively. The reaction is Zn(s) + Cu$$^{2+}$$(aq) $$\to$$ Zn$$^{2+}$$(aq) + Cu(s). The standard reaction enthalpy $$(\Delta_r H^-)$$ at 300 K in mol$$^{-1}$$ is [Use R = 8 J K$$^{-1}$$ mol$$^{-1}$$ and F = 96,500 C mol$$^{-1}$$]
JEE Main 12th January 2019 Shift 1 - Question 48
Decomposition of X exhibits a rate constant of 0.05 $$\mu$$g/year. How many years are required for the decomposition of 5 $$\mu$$g of X into 2.5 $$\mu$$g?
JEE Main 12th January 2019 Shift 1 - Question 49
In the Hall-Heroult process, aluminium is formed at the cathode. The cathode is made out of:
JEE Main 12th January 2019 Shift 1 - Question 50
Iodine reacts with concentrated $$HNO_3$$ to yield Y along with other products. The oxidation state of iodine in Y, is:


