JEE Main 12th January 2019 Shift 1
For the following questions answer them individually
JEE Main 12th January 2019 Shift 1 - Question 31
What is the work function of the metal if the light of wavelength 4000 $$\mathring{A}$$ generates photoelectrons of velocity $$6 \times 10^5$$ ms$$^{-1}$$ from it? (Mass of electron $$= 9 \times 10^{-31}$$ kg, velocity of light $$= 3 \times 10^8$$ ms$$^{-1}$$, Planck's constant $$= 6.626 \times 10^{-34}$$ Js, Charge of electron $$= 6.626 \times 10^{-34}$$ Js)
JEE Main 12th January 2019 Shift 1 - Question 32
The element with Z = 120 (not yet discovered) will be a/an
JEE Main 12th January 2019 Shift 1 - Question 33
Given
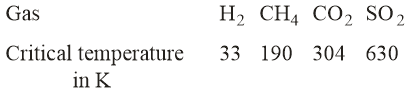
On the basis of data given above, predict which of the following gases shows the least adsorption on a definite amount of charcoal?
JEE Main 12th January 2019 Shift 1 - Question 34
The volume of gas A is twice than that of gas B. The compressibility factor of gas A is thrice than that of gas B at same temperature. What are the pressures of the gases for equal number of moles?
JEE Main 12th January 2019 Shift 1 - Question 35
For a diatomic ideal gas in a closed system, which of the following plots does not correctly describe the relation between various thermodynamic quantities?
JEE Main 12th January 2019 Shift 1 - Question 36
In a chemical reaction, $$A + 2B \xrightleftharpoons{K} 2C + D$$, the initial concentration of B was 1.5 times the concentration of A, but the equilibrium concentrations of A and B were found to be equal. The equilibrium constant (K) for the chemical reaction is:
JEE Main 12th January 2019 Shift 1 - Question 37
Two solids dissociate as follows: $$A(s) \rightleftharpoons B(g) + C(g)$$; $$K_{P_1} = x$$ atm$$^2$$, $$D(s) \rightleftharpoons C(g) + E(g)$$; $$K_{P_2} = y$$ atm$$^2$$. The total pressure when both the solids dissociate simultaneously is:
JEE Main 12th January 2019 Shift 1 - Question 38
50 mL of 0.5 M oxalic acid is needed to neutralize 25 mL of sodium hydroxide solution. What is the amount of NaOH in 50 mL of the given sodium hydroxide solution?
JEE Main 12th January 2019 Shift 1 - Question 39
What is the hardness of a water sample (in terms of equivalents of CaCO$$_3$$) containing $$10^{-3}$$ M CaSO$$_4$$? (Molar mass of CaSO$$_4$$ = 136 g mol$$^{-1}$$)
JEE Main 12th January 2019 Shift 1 - Question 40
A metal on combustion in excess air forms X. X upon hydrolysis with water yields $$H_2O_2$$ and $$O_2$$ along with another product. The metal is:


