NTA JEE Main 12th January 2019 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Main 12th January 2019 Shift 1 - Chemistry - Question 31
What is the work function of the metal if the light of wavelength 4000 $$\mathring{A}$$ generates photoelectrons of velocity $$6 \times 10^5$$ ms$$^{-1}$$ from it? (Mass of electron $$= 9 \times 10^{-31}$$ kg, velocity of light $$= 3 \times 10^8$$ ms$$^{-1}$$, Planck's constant $$= 6.626 \times 10^{-34}$$ Js, Charge of electron $$= 6.626 \times 10^{-34}$$ Js)
NTA JEE Main 12th January 2019 Shift 1 - Chemistry - Question 32
The element with Z = 120 (not yet discovered) will be a/an
NTA JEE Main 12th January 2019 Shift 1 - Chemistry - Question 33
Given
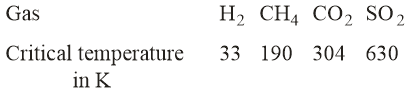
On the basis of data given above, predict which of the following gases shows the least adsorption on a definite amount of charcoal?
NTA JEE Main 12th January 2019 Shift 1 - Chemistry - Question 34
The volume of gas A is twice than that of gas B. The compressibility factor of gas A is thrice than that of gas B at same temperature. What are the pressures of the gases for equal number of moles?
NTA JEE Main 12th January 2019 Shift 1 - Chemistry - Question 35
For a diatomic ideal gas in a closed system, which of the following plots does not correctly describe the relation between various thermodynamic quantities?
NTA JEE Main 12th January 2019 Shift 1 - Chemistry - Question 36
In a chemical reaction, $$A + 2B \xrightleftharpoons{K} 2C + D$$, the initial concentration of B was 1.5 times the concentration of A, but the equilibrium concentrations of A and B were found to be equal. The equilibrium constant (K) for the chemical reaction is:
NTA JEE Main 12th January 2019 Shift 1 - Chemistry - Question 37
Two solids dissociate as follows: $$A(s) \rightleftharpoons B(g) + C(g)$$; $$K_{P_1} = x$$ atm$$^2$$, $$D(s) \rightleftharpoons C(g) + E(g)$$; $$K_{P_2} = y$$ atm$$^2$$. The total pressure when both the solids dissociate simultaneously is:
NTA JEE Main 12th January 2019 Shift 1 - Chemistry - Question 38
50 mL of 0.5 M oxalic acid is needed to neutralize 25 mL of sodium hydroxide solution. What is the amount of NaOH in 50 mL of the given sodium hydroxide solution?
NTA JEE Main 12th January 2019 Shift 1 - Chemistry - Question 39
What is the hardness of a water sample (in terms of equivalents of CaCO$$_3$$) containing $$10^{-3}$$ M CaSO$$_4$$? (Molar mass of CaSO$$_4$$ = 136 g mol$$^{-1}$$)
NTA JEE Main 12th January 2019 Shift 1 - Chemistry - Question 40
A metal on combustion in excess air forms X. X upon hydrolysis with water yields $$H_2O_2$$ and $$O_2$$ along with another product. The metal is:
NTA JEE Main 12th January 2019 Shift 1 - Chemistry - Question 41
Among the following four aromatic compounds, which one will have the lowest melting point?
NTA JEE Main 12th January 2019 Shift 1 - Chemistry - Question 42
The correct order for acid strength of compounds $$CH \equiv CH$$, $$CH_3 - C \equiv CH$$ and $$CH_2 = CH_2$$ is as follows:
NTA JEE Main 12th January 2019 Shift 1 - Chemistry - Question 43
The major product of the following reaction is:
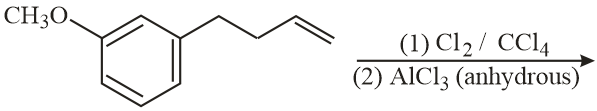
NTA JEE Main 12th January 2019 Shift 1 - Chemistry - Question 44
Water samples with BOD values of 4 ppm and 18 ppm, respectively, are:
NTA JEE Main 12th January 2019 Shift 1 - Chemistry - Question 45
The molecule that has minimum or no role in the formation of photochemical smog, is:
NTA JEE Main 12th January 2019 Shift 1 - Chemistry - Question 46
The freezing point of a 4% aqueous solution of X is equal to the freezing point of a 12% aqueous solution of Y. If the molecular weight of X is A, then the molecular weight of Y will be
NTA JEE Main 12th January 2019 Shift 1 - Chemistry - Question 47
The standard electrode potential E° and its temperature coefficient $$\left(\frac{dE}{dT}\right)$$ for a cell are 2 V and $$-5 \times 10^{-4}$$ V K$$^{-1}$$ at 300 K, respectively. The reaction is Zn(s) + Cu$$^{2+}$$(aq) $$\to$$ Zn$$^{2+}$$(aq) + Cu(s). The standard reaction enthalpy $$(\Delta_r H^-)$$ at 300 K in mol$$^{-1}$$ is [Use R = 8 J K$$^{-1}$$ mol$$^{-1}$$ and F = 96,500 C mol$$^{-1}$$]
NTA JEE Main 12th January 2019 Shift 1 - Chemistry - Question 48
Decomposition of X exhibits a rate constant of 0.05 $$\mu$$g/year. How many years are required for the decomposition of 5 $$\mu$$g of X into 2.5 $$\mu$$g?
NTA JEE Main 12th January 2019 Shift 1 - Chemistry - Question 49
In the Hall-Heroult process, aluminium is formed at the cathode. The cathode is made out of:
NTA JEE Main 12th January 2019 Shift 1 - Chemistry - Question 50
Iodine reacts with concentrated $$HNO_3$$ to yield Y along with other products. The oxidation state of iodine in Y, is:
NTA JEE Main 12th January 2019 Shift 1 - Chemistry - Question 51
The pair of metal ions that can give a spin only magnetic moment of 3.9 BM for the complex $$[M(H_2O)_6]Cl_2$$, is:
NTA JEE Main 12th January 2019 Shift 1 - Chemistry - Question 52
The metal's d-orbitals that are directly facing the ligands in $$K_3[Co(CN)_6]$$ are:
NTA JEE Main 12th January 2019 Shift 1 - Chemistry - Question 53
$$Mn_2(CO)_{10}$$ is an organometallic compound due to the presence of:
NTA JEE Main 12th January 2019 Shift 1 - Chemistry - Question 54
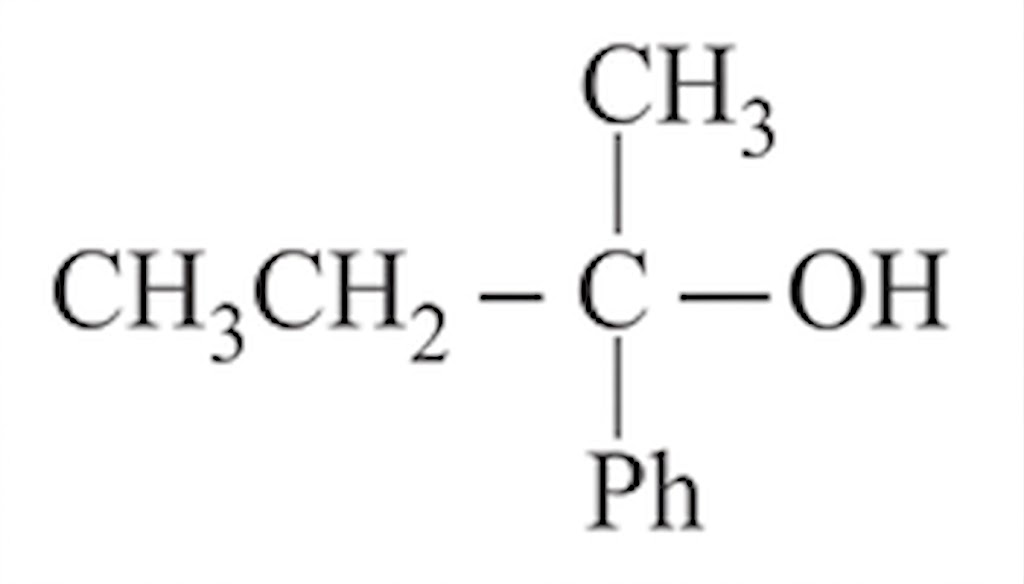
cannot be prepared by:
NTA JEE Main 12th January 2019 Shift 1 - Chemistry - Question 55
In the following reactions, products A and B are:
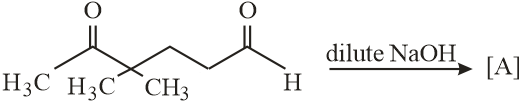
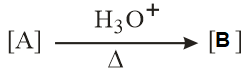
NTA JEE Main 12th January 2019 Shift 1 - Chemistry - Question 56
The major product of the following reaction is:
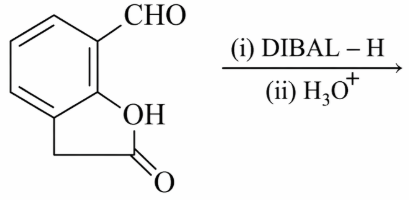
NTA JEE Main 12th January 2019 Shift 1 - Chemistry - Question 57
In the following reaction:
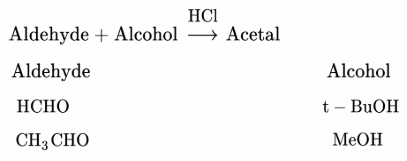
The best combination is:
NTA JEE Main 12th January 2019 Shift 1 - Chemistry - Question 58
The increasing order of reactivity of the following compounds towards reaction with alkyl halides directly is:
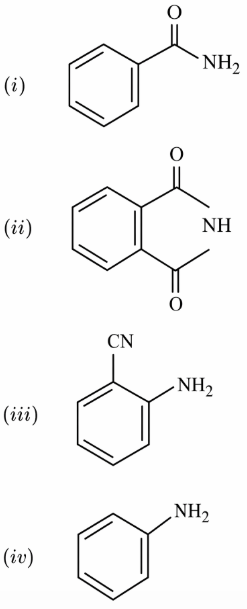
NTA JEE Main 12th January 2019 Shift 1 - Chemistry - Question 59
Poly-$$\beta$$-hydroxybutyrate-co-$$\beta$$-hydroxyvalerate (PHBV) is a copolymer of ____.
NTA JEE Main 12th January 2019 Shift 1 - Chemistry - Question 60
Among the following compounds most basic amino acid is:
.webp)
.webp)
.webp)
.webp)




