NTA JEE Main 11th January 2019 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Main 11th January 2019 Shift 1 - Chemistry - Question 31
A 10 mg effervescent tablet containing sodium bicarbonate and oxalic acid releases 0.25 mL of $$CO_2$$ at T = 298.15 K and P = 1 bar. If molar volume of $$CO_2$$ is 25.0 L under such condition, what is the percentage of sodium bicarbonate in each tablet? [Molar mass of $$NaHCO_3 = 84$$ g $$mol^{-1}$$]
NTA JEE Main 11th January 2019 Shift 1 - Chemistry - Question 32
Heat treatment of muscular pain involves radiation of wavelength of about 900 nm. Which spectral line of H atom is suitable for this purpose? [$$R_H = 1 \times 10^5 \text{ cm}^{-1}$$, $$h = 6.6 \times 10^{-34}$$ Js, $$c = 3 \times 10^8 \text{ ms}^{-1}$$]
NTA JEE Main 11th January 2019 Shift 1 - Chemistry - Question 33
The correct order of the atomic radii of C, Cs, Al, and S is:
NTA JEE Main 11th January 2019 Shift 1 - Chemistry - Question 34
Two blocks of the same metal having same mass and at temperature $$T_1$$, and $$T_2$$, respectively, are brought in contact with each other and allowed to attain thermal equilibrium at constant pressure. The change in entropy, $$\Delta S$$, for this process is:
NTA JEE Main 11th January 2019 Shift 1 - Chemistry - Question 35
For the chemical reaction $$X \rightleftharpoons Y$$, the standard reaction Gibbs energy depends on temperature T (in K) as $$\Delta_r G°$$ (in kJ mol$$^{-1}$$) $$= 120 - \frac{3}{8}T$$. The major component of the reaction mixture at T is:
NTA JEE Main 11th January 2019 Shift 1 - Chemistry - Question 36
Consider the reaction $$N_2(g) + 3H_2(g) \rightleftharpoons 2NH_3(g)$$. The equilibrium constant of the above reaction is $$K_P$$. If pure ammonia is left to dissociate, the partial pressure of ammonia at equilibrium is given by (Assume that $$P_{NH_3} \ll P_{total}$$ at equilibrium)
NTA JEE Main 11th January 2019 Shift 1 - Chemistry - Question 37
The amphoteric hydroxide is:
NTA JEE Main 11th January 2019 Shift 1 - Chemistry - Question 38
The correct statements among (a) to (d) regarding $$H_2$$ as a fuel are: (i) It produces less pollutants than petrol. (ii) A cylinder of compressed dihydrogen weighs ~ 30 times more than a petrol tank producing the same amount of energy. (iii) Dihydrogen is stored in tanks of metal alloys like $$NaNi_5$$. (iv) On combustion, values of energy released per gram of liquid dihydrogen and LPG are 50 and 142 kJ, respectively.
NTA JEE Main 11th January 2019 Shift 1 - Chemistry - Question 39
NaH is an example of:
NTA JEE Main 11th January 2019 Shift 1 - Chemistry - Question 40
Which compound(s) out of the following is/are not aromatic?
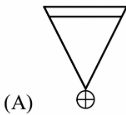
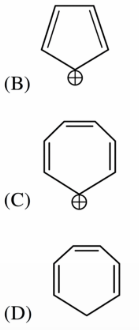
NTA JEE Main 11th January 2019 Shift 1 - Chemistry - Question 41
The correct match between items I and II is:

NTA JEE Main 11th January 2019 Shift 1 - Chemistry - Question 42
An organic compound is estimated through Dumas method and was found to evolve 6 moles of $$CO_2$$, 4 moles of $$H_2O$$ and 1 mole of nitrogen gas. The formula of the compound is:
NTA JEE Main 11th January 2019 Shift 1 - Chemistry - Question 43
Peroxyacetyl nitrate (PAN), an eye irritant, is produced by:
NTA JEE Main 11th January 2019 Shift 1 - Chemistry - Question 44
The concentration of dissolved oxygen (DO) in cold water can go upto
NTA JEE Main 11th January 2019 Shift 1 - Chemistry - Question 45
A solid having density of $$9 \times 10^3$$ kg m$$^{-3}$$ forms face centred cubic crystals of edge length $$200\sqrt{2}$$ pm. What is the molar mass of the solid? [Avogadro constant $$\approx 6 \times 10^{23}$$ mol$$^{-1}$$, $$\pi \approx 3$$]
NTA JEE Main 11th January 2019 Shift 1 - Chemistry - Question 46
The freezing point of a diluted milk sample is found to be $$-0.2°C$$, while it should have been $$-0.5°C$$ for pure milk. How much water has been added to pure milk to make the diluted sample?
NTA JEE Main 11th January 2019 Shift 1 - Chemistry - Question 47
For the cell $$Zn(s) | Zn^{2+}(aq) || M^{x+}(aq) | M(s)$$, different half cells and their standard electrode potentials are given below:
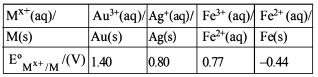
If $$E°_{Zn^{2+}/Zn} = -0.76$$ V, which cathode will give a maximum value of $$E°_{cell}$$ per electron transferred?
NTA JEE Main 11th January 2019 Shift 1 - Chemistry - Question 48
If a reaction follows the Arrhenius equation, the plot $$\ln k$$ vs $$\frac{1}{(RT)}$$ gives straight line with a gradient $$(-y)$$ unit. The energy required to activate the reactant is:
NTA JEE Main 11th January 2019 Shift 1 - Chemistry - Question 49
An example of solid sol is:
NTA JEE Main 11th January 2019 Shift 1 - Chemistry - Question 50
Match the ores (column A) with the metals (column B):

NTA JEE Main 11th January 2019 Shift 1 - Chemistry - Question 51
The chloride that CANNOT get hydrolysed is:
NTA JEE Main 11th January 2019 Shift 1 - Chemistry - Question 52
The element that usually does NOT show variable oxidation states is:
NTA JEE Main 11th January 2019 Shift 1 - Chemistry - Question 53
The major product of the following reaction is:
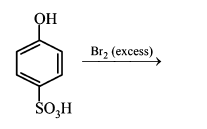
NTA JEE Main 11th January 2019 Shift 1 - Chemistry - Question 54
The major product of the following reaction is:
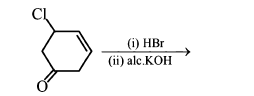
NTA JEE Main 11th January 2019 Shift 1 - Chemistry - Question 55
The major product of the following reaction is:
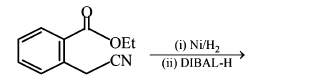
NTA JEE Main 11th January 2019 Shift 1 - Chemistry - Question 56
The major product of the following reaction is:
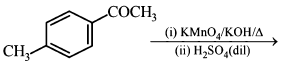
NTA JEE Main 11th January 2019 Shift 1 - Chemistry - Question 57
The polymer obtained from the following reactions is:
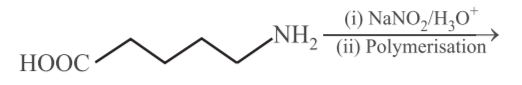
NTA JEE Main 11th January 2019 Shift 1 - Chemistry - Question 58
The correct match between item (I) and item (II) is:

NTA JEE Main 11th January 2019 Shift 1 - Chemistry - Question 59
Match the metals (column I) with the coordination compound(s)/enzyme(s) (column II):

NTA JEE Main 11th January 2019 Shift 1 - Chemistry - Question 60
Among the following compounds, which one is found in RNA?
.webp)
.webp)
.webp)
.webp)




