NTA JEE Main 11th April 2023 Shift 1
For the following questions answer them individually
NTA JEE Main 11th April 2023 Shift 1 - Question 41
In the extraction process of copper, the product obtained after carrying out the reactions
(i) $$2Cu_2S + 3O_2 \to 2Cu_2O + 2SO_2$$
(ii) $$2Cu_2O + Cu_2S \to 6Cu + SO_2$$ is called
NTA JEE Main 11th April 2023 Shift 1 - Question 42
Given below are two statements:
Statement-I: Methane and steam passed over a heated Ni catalyst produces hydrogen gas.
Statement-II: Sodium nitrite reacts with NH$$_4$$Cl to give H$$_2$$O, N$$_2$$ and NaCl.
In the light of the above statements, choose the most appropriate answer from the options below:
NTA JEE Main 11th April 2023 Shift 1 - Question 43
When a solution of mixture of two inorganic salts was treated with freshly prepared ferrous sulphate in acidic medium, a dark brown ring was formed whereas on treatment with neutral FeCl$$_3$$, it gave deep red colour which disappeared on boiling and a brown red ppt was formed. The mixture contains
NTA JEE Main 11th April 2023 Shift 1 - Question 44
The complex that dissolves in water is
NTA JEE Main 11th April 2023 Shift 1 - Question 45
The set which does not have ambidentate ligand(s) is
NTA JEE Main 11th April 2023 Shift 1 - Question 46
Which of the following complex has a possibility to exist as meridional isomer?
NTA JEE Main 11th April 2023 Shift 1 - Question 47
L-isomer of tetrose X (C$$_4$$H$$_8$$O$$_4$$) gives positive Schiff's test and has two chiral carbons. On acetylation 'X' yields triacetate. 'X' also undergoes following reactions
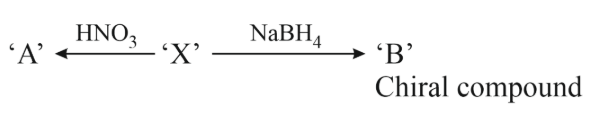
NTA JEE Main 11th April 2023 Shift 1 - Question 48
'A' and 'B' in the above reaction are :
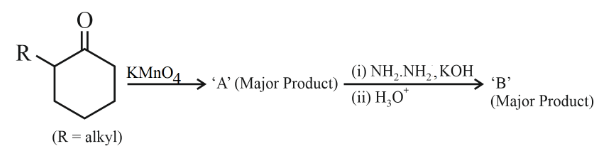
NTA JEE Main 11th April 2023 Shift 1 - Question 49
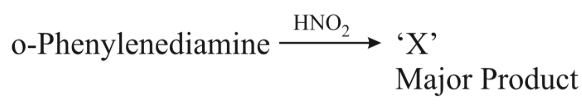
. 'X' is:
NTA JEE Main 11th April 2023 Shift 1 - Question 50
The polymer X-consists of linear molecules and is closely packed. It is prepared in the presence of trimethylaluminiumpropyl and titanium tetrachloride under low pressure. The polymer X is


