NTA JEE Main 11th April 2023 Shift 1
For the following questions answer them individually
NTA JEE Main 11th April 2023 Shift 1 - Question 31
25 mL of silver nitrate solution (1M) is added dropwise to 25 mL of potassium iodide (1.05 M) solution. The ion(s) present in very small quantity in the solution is/are
NTA JEE Main 11th April 2023 Shift 1 - Question 32
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R:
Assertion A: In the photoelectric effect, the electrons are ejected from the metal surface as soon as the beam of light of frequency greater than threshold frequency strikes the surface.
Reason R: When the photon of any energy strikes an electron in the atom, transfer of energy from the photon to the electron takes place.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Main 11th April 2023 Shift 1 - Question 33
For compound having the formula GaAlCl$$_4$$, the correct option from the following is
NTA JEE Main 11th April 2023 Shift 1 - Question 34
For elements B, C, N, Li, Be, O and F, the correct order of first ionisation enthalpy is
NTA JEE Main 11th April 2023 Shift 1 - Question 35
Match List-I with List-II:
| List-I Species | List-II Geometry/Shape | ||
|---|---|---|---|
| A. | H$$_3$$O$$^+$$ | I. | Tetrahedral |
| B. | Acetylide anion | II. | Linear |
| C. | NH$$_4^+$$ | III. | Pyramidal |
| D. | ClO$$_2^-$$ | IV. | Bent |
Choose the correct answer from the options given below:
NTA JEE Main 11th April 2023 Shift 1 - Question 36
Match List-I with List-II:
| List-I | List-II | ||
|---|---|---|---|
| A. | K | I. | Thermonuclear reactions |
| B. | KCl | II. | Fertilizer |
| C. | KOH | III. | Sodium potassium pump |
| D. | Li | IV. | Absorbent of CO$$_2$$ |
NTA JEE Main 11th April 2023 Shift 1 - Question 37
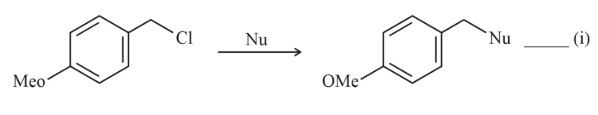
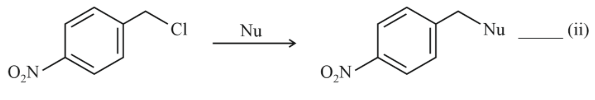
Where Nu = Nucleophile
Find out the correct statement from the options given below for the above two reactions.
NTA JEE Main 11th April 2023 Shift 1 - Question 38
Thin layer chromatography of a mixture shows the following observation.
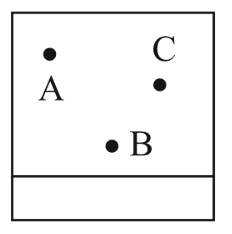
The correct order of elution in the silica gel column chromatography is
NTA JEE Main 11th April 2023 Shift 1 - Question 39
Arrange the following compounds in increasing order of rate of aromatic electrophilic substitution reaction.
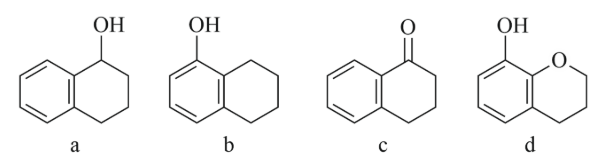
NTA JEE Main 11th April 2023 Shift 1 - Question 40
Given below are two statements:
Statement-I: If BOD is 4 ppm and dissolved oxygen is 8 ppm, then it is a good quality water.
Statement-II: If the concentration of zinc and nitrate salts are 5 ppm each, then it can be a good quality water.
In the light of the above statements, choose the most appropriate answer from the options below:


