NTA JEE Main 11th April 2023 Shift 1
For the following questions answer them individually
NTA JEE Main 11th April 2023 Shift 1 - Question 51
A solution of sugar is obtained by mixing 200 g of its 25% solution and 500 g of its 40% solution (both by mass). The mass percentage of the resulting sugar solution is _______ (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 11th April 2023 Shift 1 - Question 52
Solid fuel used in rocket is a mixture of Fe$$_2$$O$$_3$$ and Al (in ratio 1 : 2). The heat evolved (kJ) per gram of the mixture is _______
Given $$\Delta H_f^0$$ Al$$_2$$O$$_3$$ = -1700 kJ mol$$^{-1}$$
$$\Delta H_f^0$$ Fe$$_2$$O$$_3$$ = -840 kJ mol$$^{-1}$$
Molar mass of Fe, Al and O are 56, 27 and 16 g mol$$^{-1}$$ respectively
789
456
123
0.-
Clear All
NTA JEE Main 11th April 2023 Shift 1 - Question 53
A mixture of one mole of H$$_2$$O and 1 mole of CO is taken in a 10 litre container and heated to 725 K. At equilibrium 40% of water by mass reacts with carbon monoxide according to the equation:
$$CO(g) + H_2O(g) \rightleftharpoons CO_2(g) + H_2(g)$$
The equilibrium constant $$K_C \times 10^2$$ for the reaction is _______ (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 11th April 2023 Shift 1 - Question 54
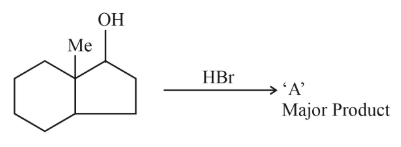
The number of hyperconjugation structures involved to stabilize carbocation formed in the reaction of 2-methylcyclohexan-1-ol with HBr is _______
789
456
123
0.-
Clear All
NTA JEE Main 11th April 2023 Shift 1 - Question 55
An atomic substance A of molar mass 12 g mol$$^{-1}$$ has a cubic crystal structure with edge length of 300 pm. The no. of atoms present in one unit cell of A is _______ (Nearest integer)
Given the density of A is 3.0 g m m$$^{-1}$$ and NA$$_A$$ = $$6.02 \times 10^{23}$$ mol$$^{-1}$$
789
456
123
0.-
Clear All
NTA JEE Main 11th April 2023 Shift 1 - Question 56
0.004 M K$$_2$$SO$$_4$$ solution is isotonic with 0.01 M glucose solution. Percentage dissociation of K$$_2$$SO$$_4$$ is _______ (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 11th April 2023 Shift 1 - Question 57
In an electrochemical reaction of lead, at standard temperature, if $$E^0_{Pb^{2+}/Pb} = m$$ Volt and $$E^0_{Pb^{4+}/Pb} = n$$ Volt, then the value of $$E^0Pb^{2+}/Pb^{4+}$$ is given by $$m - xn$$. The value of x is _______ (Nearest integer).
789
456
123
0.-
Clear All
NTA JEE Main 11th April 2023 Shift 1 - Question 58
KClO$$_3$$ + 6FeSO$$_4$$ + 3H$$_2$$SO$$_4$$ $$\to$$ KCl + 3Fe$$_2$$(SO$$_4$$)$$_3$$ + 3H$$_2$$O
The above reaction was studied at 300 K by monitoring the concentration of FeSO$$_4$$ in which initial concentration was 10 M and after half an hour became 8.8 M. The rate of production of Fe$$_2$$(SO$$_4$$)$$_3$$ is _______ $$\times 10^{-6}$$ mol L$$^{-1}$$ s$$^{-1}$$ (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 11th April 2023 Shift 1 - Question 59
The ratio of spin-only magnetic moment values $$\mu_{eff}[Cr(CN)_6]^{3-}$$ / $$\mu_{eff}[Cr(H_2O)_6]^{3+}$$ is _______
789
456
123
0.-
Clear All
NTA JEE Main 11th April 2023 Shift 1 - Question 60
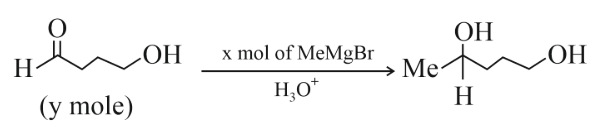
The ratio x/y on completion of the reaction is
789
456
123
0.-
Clear All


