JEE Advanced 2025 Paper-1
For the following questions answer them individually
JEE Advanced 2025 Paper-1 - Question 41
At 25 $$^\circ$$C, the concentration of H$$^+$$ ions in $$1.00 \times 10^{-3}$$ M aqueous solution of a weak monobasic acid having acid dissociation constant ($$K_a$$) of $$4.00 \times 10^{-11}$$ is $$X \times 10^{-7}$$ M. The value of $$X$$ is ________.
Use: Ionic product of water ($$K_w$$) = $$1.00 \times 10^{-14}$$ at 25 $$^\circ$$C
789
456
123
0.-
Clear All
JEE Advanced 2025 Paper-1 - Question 42
Molar volume ($$V_m$$) of a van der Waals gas can be calculated by expressing the van der Waals equation as a cubic equation with $$V_m$$ as the variable. The ratio (in mol dm$$^{-3}$$) of the coefficient of $$V_m^2$$ to the coefficient of $$V_m$$ for a gas having van der Waals constants $$a = 6.0$$ dm$$^6$$ atm mol$$^{-2}$$ and $$b = 0.060$$ dm$$^3$$ mol$$^{-1}$$ at 300 K and 300 atm is ________.
Use: Universal gas constant (R) = 0.082 dm$$^3$$ atm mol$$^{-1}$$ K$$^{-1}$$
789
456
123
0.-
Clear All
JEE Advanced 2025 Paper-1 - Question 43
Considering ideal gas behavior, the expansion work done (in kJ) when 144 g of water is electrolyzed completely under constant pressure at 300 K is ________.
Use: Universal gas constant (R) = 8.3 J K$$^{-1}$$ mol$$^{-1}$$; Atomic mass (in amu): H = 1, O = 16
789
456
123
0.-
Clear All
JEE Advanced 2025 Paper-1 - Question 44
The monomer (X) involved in the synthesis of Nylon 6,6 gives positive carbylamine test. If 10 moles of X are analyzed using Dumas method, the amount (in grams) of nitrogen gas evolved is ________.
Use: Atomic mass of N (in amu) = 14
789
456
123
0.-
Clear All
JEE Advanced 2025 Paper-1 - Question 45
The reaction sequence given below is carried out with 16 moles of X. The yield of the major product in each step is given below the product in parentheses. The amount (in grams) of S produced is ________.
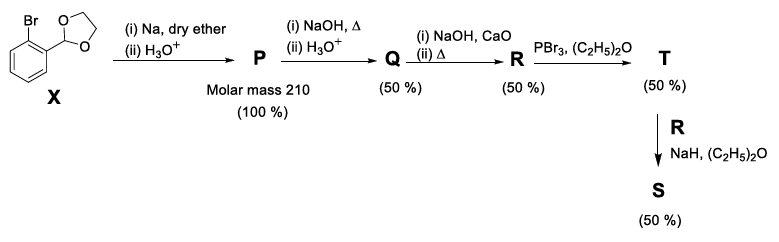
Use: Atomic mass (in amu): H = 1, C = 12, O = 16, Br = 80
789
456
123
0.-
Clear All
JEE Advanced 2025 Paper-1 - Question 46
The correct match of the group reagents in List-I for precipitating the metal ion given in List-II from solutions, is

JEE Advanced 2025 Paper-1 - Question 47
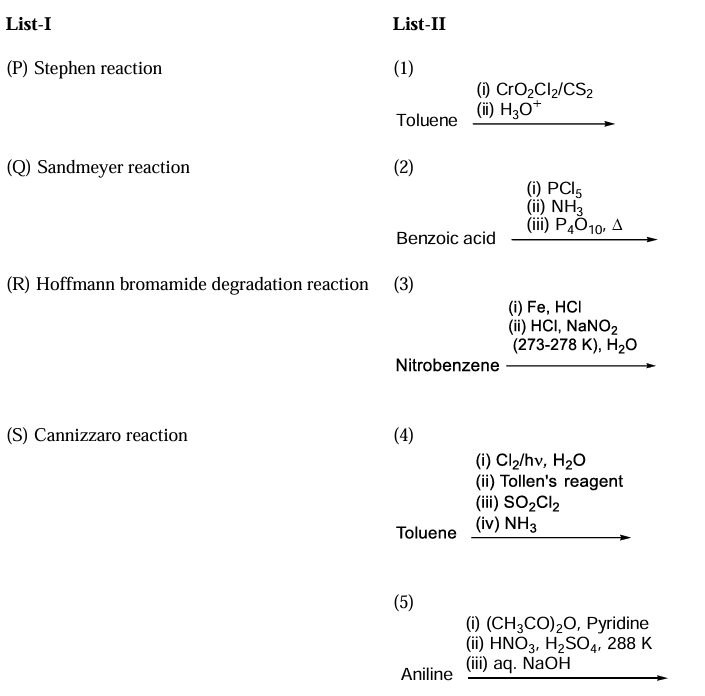
The major products obtained from the reactions in List-II are the reactants for the named reactions mentioned in List-I. Match each entry in List-I with the appropriate entry in List-II and choose the correct option.
JEE Advanced 2025 Paper-1 - Question 48
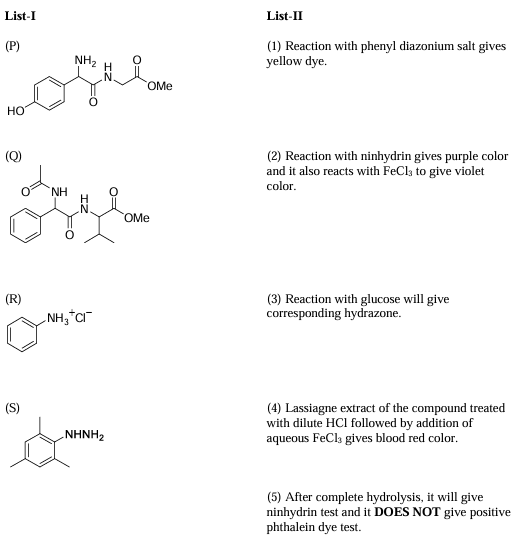
Match the compounds in List-I with the appropriate observations in List-II and choose the correct option.