JEE Advanced 2025 Paper-1
For the following questions answer them individually
JEE Advanced 2025 Paper-1 - Question 31
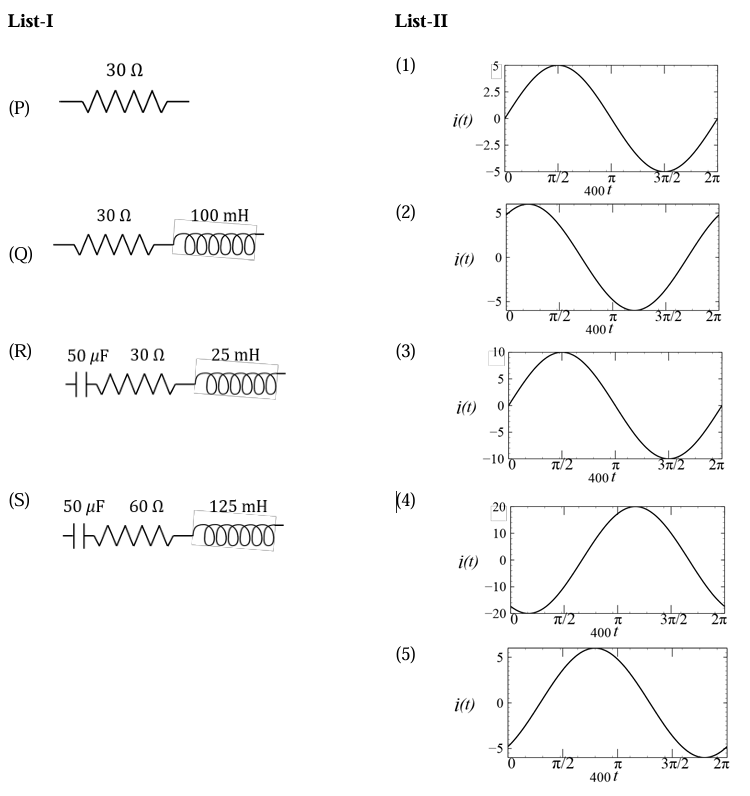
A circuit with an electrical load having impedance $$Z$$ is connected with an AC source as shown in the diagram. The source voltage varies in time as $$V(t) = 300 \sin(400t)$$ V, where $$t$$ is time in s. List-I shows various options for the load. The possible currents $$i(t)$$ in the circuit as a function of time are given in List-II.
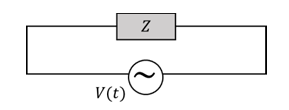
Choose the option that describes the correct match between the entries in List-I to those in List-II.
JEE Advanced 2025 Paper-1 - Question 32
List-I shows various functional dependencies of energy ($$E$$) on the atomic number ($$Z$$). Energies associated with certain phenomena are given in List-II.
Choose the option that describes the correct match between the entries in List-I to those in List-II.

JEE Advanced 2025 Paper-1 - Question 33
The heating of NH$$_4$$NO$$_2$$ at 60-70 $$^\circ$$C and NH$$_4$$NO$$_3$$ at 200-250 $$^\circ$$C is associated with the formation of nitrogen containing compounds X and Y, respectively. X and Y, respectively, are
JEE Advanced 2025 Paper-1 - Question 34
The correct order of the wavelength maxima of the absorption band in the ultraviolet-visible region for the given complexes is
JEE Advanced 2025 Paper-1 - Question 35
One of the products formed from the reaction of permanganate ion with iodide ion in neutral aqueous medium is
JEE Advanced 2025 Paper-1 - Question 36
Consider the depicted hydrogen (H) in the hydrocarbons given below. The most acidic hydrogen (H) is
JEE Advanced 2025 Paper-1 - Question 37
Regarding the molecular orbital (MO) energy levels for homonuclear diatomic molecules, the INCORRECT statement(s) is(are)
JEE Advanced 2025 Paper-1 - Question 38
The pair(s) of diamagnetic ions is(are)
JEE Advanced 2025 Paper-1 - Question 39
For the reaction sequence given below, the correct statement(s) is(are)
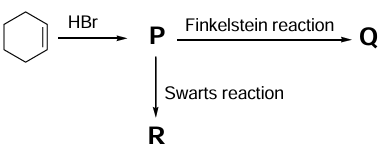
(In the options, X is any atom other than carbon and hydrogen, and it is different in P, Q and R)
JEE Advanced 2025 Paper-1 - Question 40
In an electrochemical cell, dichromate ions in aqueous acidic medium are reduced to Cr$$^{3+}$$. The current (in amperes) that flows through the cell for 48.25 minutes to produce 1 mole of Cr$$^{3+}$$ is ________.
Use: 1 Faraday = 96500 C mol$$^{-1}$$
789
456
123
0.-
Clear All



