JEE Advanced 2023 Paper-2
For the following questions answer them individually
JEE Advanced 2023 Paper-2 - Question 41
In the following reactions, P, Q, R, and S are the major products.
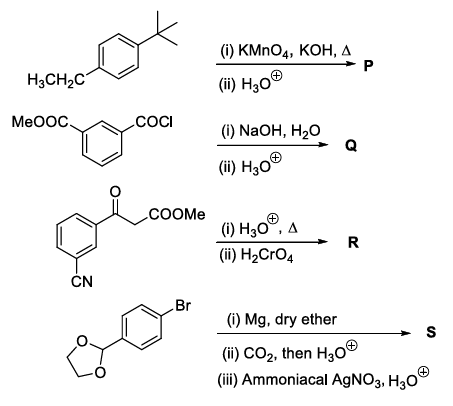
The correct statement(s) about P, Q, R, and S is (are)
JEE Advanced 2023 Paper-2 - Question 42
H$$_2$$S (5 moles) reacts completely with acidified aqueous potassium permanganate solution. In this reaction, the number of moles of water produced is x, and the number of moles of electrons involved is y. The value of (x + y) is ____.
789
456
123
0.-
Clear All
JEE Advanced 2023 Paper-2 - Question 43
Among [I$$_3$$]$$^+$$, [SiO$$_4$$]$$^{4-}$$, SO$$_2$$Cl$$_2$$, XeF$$_2$$, SF$$_4$$, ClF$$_3$$, Ni(CO)$$_4$$, XeO$$_2$$F$$_2$$, [PtCl$$_4$$]$$^{2-}$$, XeF$$_4$$, and SOCl$$_2$$, the total number of species having $$sp^3$$ hybridised central atom is ____.
789
456
123
0.-
Clear All
JEE Advanced 2023 Paper-2 - Question 44
Consider the following molecules: Br$$_3$$O$$_8$$, F$$_2$$O, H$$_2$$S$$_4$$O$$_6$$, H$$_2$$S$$_5$$O$$_6$$, and C$$_3$$O$$_2$$.
Count the number of atoms existing in their zero oxidation state in each molecule. Their sum is ____.
789
456
123
0.-
Clear All
JEE Advanced 2023 Paper-2 - Question 45
For He$$^+$$, a transition takes place from the orbit of radius 105.8 pm to the orbit of radius 26.45 pm. The wavelength (in nm) of the emitted photon during the transition is ____.
[Use: Bohr radius, $$a = 52.9$$ pm; Rydberg constant, $$R_H = 2.2 \times 10^{-18}$$ J; Planck's constant, $$h = 6.6 \times 10^{-34}$$ J s; Speed of light, $$c = 3 \times 10^8$$ m s$$^{-1}$$]
789
456
123
0.-
Clear All
JEE Advanced 2023 Paper-2 - Question 46
50 mL of 0.2 molal urea solution (density = 1.012 g mL$$^{-1}$$ at 300 K) is mixed with 250 mL of a solution containing 0.06 g of urea. Both the solutions were prepared in the same solvent. The osmotic pressure (in Torr) of the resulting solution at 300 K is ____.
[Use: Molar mass of urea = 60 g mol$$^{-1}$$; gas constant, R = 62 L Torr K$$^{-1}$$ mol$$^{-1}$$; Assume, $$\Delta_{mix}H = 0$$, $$\Delta_{mix}V = 0$$]
789
456
123
0.-
Clear All
JEE Advanced 2023 Paper-2 - Question 47
The reaction of 4-methyloct-1-ene (P, 2.52 g) with HBr in the presence of (C$$_6$$H$$_5$$CO)$$_2$$O$$_2$$ gives two isomeric bromides in a 9 : 1 ratio, with combined yield of 50%. Of these, the entire amount of the primary alkyl bromide was reacted with an appropriate amount of diethylamine followed by treatment with eq. K$$_2$$CO$$_3$$ to give a non-ionic product S in 100% yield.
The mass (in mg) of S obtained is ____.
[Use molar mass (in g mol$$^{-1}$$): H = 1, C = 12, N = 14, Br = 80]
789
456
123
0.-
Clear All
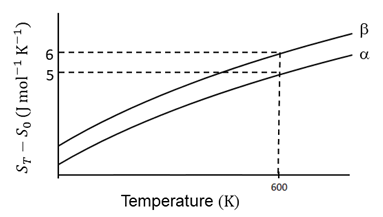
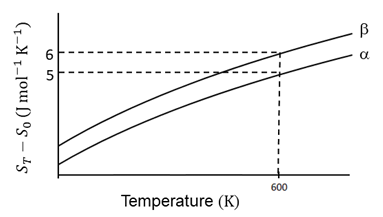
The entropy versus temperature plot for phases $$\alpha$$ and $$\beta$$ at 1 bar pressure is given. $$S_T$$ and $$S_0$$ are entropies of the phases at temperatures T and 0 K, respectively.
The transition temperature for $$\alpha$$ to $$\beta$$ phase change is 600 K and $$C_{P,\beta} - C_{P,\alpha} = 1$$ J mol$$^{-1}$$ K$$^{-1}$$. Assume $$(C_{P,\beta} - C_{P,\alpha})$$ is independent of temperature in the range of 200 to 700 K. $$C_{P,\alpha}$$ and $$C_{P,\beta}$$ are heat capacities of $$\alpha$$ and $$\beta$$ phases, respectively.
JEE Advanced 2023 Paper-2 - Question 48
The value of entropy change, $$S_\beta - S_\alpha$$ (in J mol$$^{-1}$$ K$$^{-1}$$), at 300 K is ____.
[Use: ln 2 = 0.69]
[Given: $$S_\beta - S_\alpha = 0$$ at 0 K]
789
456
123
0.-
Clear All
JEE Advanced 2023 Paper-2 - Question 49
The value of enthalpy change, $$H_\beta - H_\alpha$$ (in J mol$$^{-1}$$), at 300 K is ____.
789
456
123
0.-
Clear All
A trinitro compound, 1, 3, 5 tris-(4-nitrophenyl) benzene, on complete reaction with an excess of Sn/HCl gives major product, which on treatment with an excess of NaNO$$_2$$/HCl at 0°C provides P as the product. P, upon treatment with excess of H$$_2$$O at room temperature, gives the product Q. Bromination of Q in aqueous medium furnishes the product R. The compound P upon treatment with an excess of phenol under basic conditions gives the product S.
The molar mass difference between compounds Q and R is 474 g mol$$^{-1}$$ and between compounds P and S is 172.5 g mol$$^{-1}$$.
JEE Advanced 2023 Paper-2 - Question 50
The number of heteroatoms present in one molecule of R is ____.
[Use: Molar mass (in g mol$$^{-1}$$): H = 1, C = 12, N = 14, O = 16, Br = 80, Cl = 35.5
Atoms other than C and H are considered as heteroatoms]
789
456
123
0.-
Clear All



