JEE Advanced 2022 Paper-1
Instructions
For the following questions answer them individually
JEE Advanced 2022 Paper-1 - Question 51
Match the rate expressions in LIST-I for the decomposition of X with the corresponding profiles provided in LIST-II. $$X_s$$ and k constants having appropriate units.
| LIST-I | LIST-II |
|---|---|
| (I) rate = $$\frac{k[X]}{X_s + [X]}$$, under all possible initial concentration of X | (P) 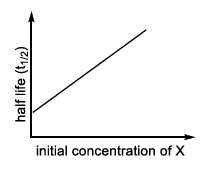 |
| (II) rate = $$\frac{k[X]}{X_s + [X]}$$, where initial concentration of X are much less than $$X_s$$ | (Q) 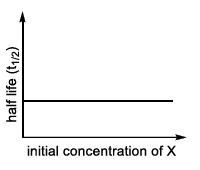 |
| (III) rate = $$\frac{k[X]}{X_s + [X]}$$, where initial concentration of X are much higher than $$X_s$$ | (R) 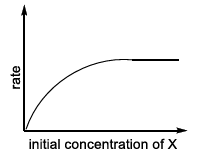 |
| (IV) rate = $$\frac{k[X]^2}{X_s + [X]}$$, where initial concentration of X is much higher than $$X_s$$ | (S)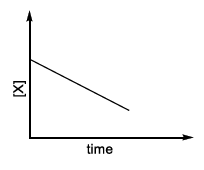 |
(T) 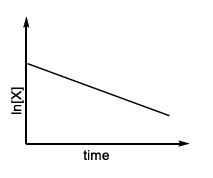 |
JEE Advanced 2022 Paper-1 - Question 52
LIST-I contains compounds and LIST-II contains reaction
| LIST-I | LIST-II |
|---|---|
| (I) H$$_2$$O$$_2$$ | (P) Mg(HCO$$_3$$)$$_2$$ + Ca(OH)$$_2$$ $$\to$$ |
| (II) Mg(OH)$$_2$$ | (Q) BaO$$_2$$ + H$$_2$$SO$$_4$$ $$\to$$ |
| (III) BaCl$$_2$$ | (R) Ca(OH)$$_2$$ + MgCl$$_2$$ $$\to$$ |
| (IV) CaCO$$_3$$ | (S) BaO$$_2$$ + HCl $$\to$$ |
| (T) Ca(HCO$$_3$$)$$_2$$ + Ca(OH)$$_2$$ $$\to$$ |
Match each compound in LIST-I with its formation reaction(s) in LIST-II, and choose the correct option
JEE Advanced 2022 Paper-1 - Question 53
LIST-I contains metal species and LIST-II contains their properties.
| LIST-I | LIST-II |
|---|---|
| (I) $$[Cr(CN)_6]^{4-}$$ | (P) $$t_{2g}$$ orbitals contain 4 electrons |
| (II) $$[RuCl_6]^{2-}$$ | (Q) $$\mu$$(spin-only) = 4.9 BM |
| (III) $$[Cr(H_2O)_6]^{2+}$$ | (R) low spin complex ion |
| (IV) $$[Fe(H_2O)_6]^{2+}$$ | (S) metal ion in 4+ oxidation state |
| (T) $$d^4$$ species |
[Given : Atomic number of Cr = 24, Ru = 44, Fe = 26]
Match each metal species in LIST-I with their properties in LIST-II, and choose the correct option
JEE Advanced 2022 Paper-1 - Question 54
Match the compounds in LIST-I with the observation in LIST-II, and choose the correct option.
| LIST-I | LIST-II |
|---|---|
| (I) Aniline | (P) Sodium fusion extract of the compound on boiling with FeSO$$_4$$, followed by acidification with conc. H$$_2$$SO$$_4$$, gives Prussian blue color. |
| (II) o-Cresol | (Q) Sodium fusion extract of the compound on treatment with sodium nitroprusside gives blood red color. |
| (III) Cysteine | (R) Addition of the compound to a saturated solution of NaHCO$$_3$$ results in effervescence. |
| (IV) Caprolactam | (S) The compound reacts with bromine water to give a white precipitate. |
| (T) Treating the compound with neutral FeCl$$_3$$ solution produces violet color. |



