JEE Advanced 2022 Paper-1
For the following questions answer them individually
JEE Advanced 2022 Paper-1 - Question 31
Six charges are placed around a regular hexagon of side length $$a$$ as shown in the figure. Five of them have charge $$q$$, and the remaining one has charge $$x$$. The perpendicular from each charge to the nearest hexagon side passes through the center O of the hexagon and is bisected by the side.
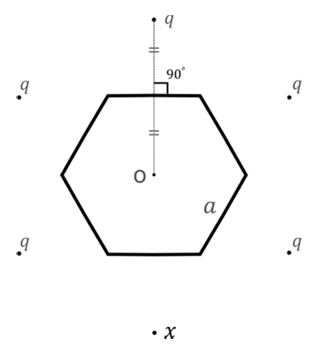
Which of the following statement(s) is(are) correct in SI units?
JEE Advanced 2022 Paper-1 - Question 32
The binding energy of nucleons in a nucleus can be affected by the pairwise Coulomb repulsion. Assume that all nucleons are uniformly distributed inside the nucleus. Let the binding energy of a proton be $$E_b^p$$ and the binding energy of a neutron be $$E_b^n$$ in the nucleus.
Which of the following statement(s) is(are) correct?
JEE Advanced 2022 Paper-1 - Question 33
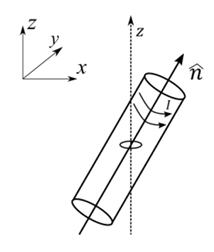
A small circular loop of area $$A$$ and resistance $$R$$ is fixed on a horizontal $$xy$$-plane with the center of the loop always on the axis $$\hat{n}$$ of a long solenoid. The solenoid has $$m$$ turns per unit length and carries current $$I$$ counterclockwise as shown in the figure. The magnetic field due to the solenoid is in $$\hat{n}$$ direction. List-I gives time dependences of $$\hat{n}$$ in terms of a constant angular frequency $$\omega$$.
List-II gives the torques experienced by the circular loop at time $$t = \frac{\pi}{6\omega}$$. Let $$\alpha = \frac{A^2 \mu_0^2 m^2 I^2 \omega}{2R}$$.
| List-I | List-II |
|---|---|
| (I) $$\frac{1}{\sqrt{2}}(\sin \omega t \, \hat{j} + \cos \omega t \, \hat{k})$$ | (P) 0 |
| (II) $$\frac{1}{\sqrt{2}}(\sin \omega t \, \hat{i} + \cos \omega t \, \hat{j})$$ | (Q) $$-\frac{\alpha}{4}\hat{i}$$ |
| (III) $$\frac{1}{\sqrt{2}}(\sin \omega t \, \hat{i} + \cos \omega t \, \hat{k})$$ | (R) $$\frac{3\alpha}{4}\hat{i}$$ |
| (IV) $$\frac{1}{\sqrt{2}}(\cos \omega t \, \hat{i} + \sin \omega t \, \hat{k})$$ | (S) $$\frac{\alpha}{4}\hat{j}$$ |
| Which one of the following options is correct? | (T) $$-\frac{3\alpha}{4}\hat{i}$$ |
JEE Advanced 2022 Paper-1 - Question 34
List I describes four systems, each with two particles $$A$$ and $$B$$ in relative motion as shown in figure. List II gives possible magnitudes of their relative velocities (in ms$$^{-1}$$) at time $$t = \frac{\pi}{3}$$ s.
| List-I | List-II |
|---|---|
(I) $$A$$ and $$B$$ are moving on a horizontal circle of radius 1 m with uniform angular speed $$\omega = 1$$ rad s$$^{-1}$$. The initial angular positions of $$A$$ and $$B$$ at time $$t = 0$$ are $$\theta = 0$$ and $$\theta = \frac{\pi}{2}$$ respectively.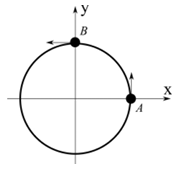 | (P) $$\frac{\sqrt{3}+1}{2}$$ |
(II) Projectiles $$A$$ and $$B$$ are fired (in the same vertical plane) at $$t = 0$$ and $$t = 0.1$$ s respectively, with the same speed $$v = \frac{5\pi}{\sqrt{2}}$$ m s$$^{-1}$$ and at $$45^\circ$$ from the horizontal plane. The initial separation between $$A$$ and $$B$$ is large enough so that they do not collide. ($$g = 10$$ m s$$^{-2}$$).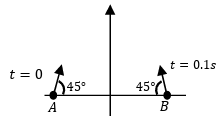 | (Q) $$\frac{\sqrt{3}-1}{\sqrt{2}}$$ |
(III) Two harmonic oscillators $$A$$ and $$B$$ moving in the $$x$$ direction according to $$x_A = x_0 \sin \frac{t}{t_0}$$ and $$x_B = x_0 \sin\left(\frac{t}{t_0} + \frac{\pi}{2}\right)$$ respectively, starting from $$t = 0$$. Take $$x_0 = 1$$ m, $$t_0 = 1$$ s.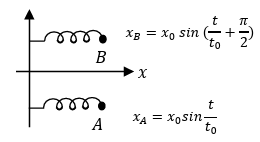 | (R) $$\sqrt{10}$$ |
(IV) Particle $$A$$ is rotating in a horizontal circular path of radius 1 m on the $$xy$$ plane, with constant angular speed $$\omega = 1$$ rad s$$^{-1}$$. Particle $$B$$ is moving up at a constant speed 3 m s$$^{-1}$$ in the vertical direction as shown in the figure. (Ignore gravity.)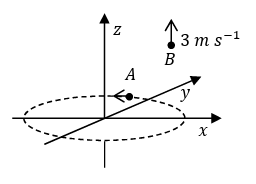 | (S) $$\sqrt{2}$$ |
| (T) $$\sqrt{25\pi^2 + 1}$$ |
Which one of the following options is correct?
JEE Advanced 2022 Paper-1 - Question 35
List I describes thermodynamic processes in four different systems. List II gives the magnitudes (either exactly or as a close approximation) of possible changes in the internal energy of the system due to the process.
| List-I | List-II |
|---|---|
| (I) $$10^{-3}$$ kg of water at 100°C is converted to steam at the same temperature, at a pressure of $$10^5$$ Pa. The volume of the system changes from $$10^{-6}$$ m$$^3$$ to $$10^{-3}$$ m$$^3$$ in the process. Latent heat of water = 2250 kJ/kg. | (P) 2 kJ |
| (II) 0.2 moles of a rigid diatomic ideal gas with volume $$V$$ at temperature 500 K undergoes an isobaric expansion to volume 3V. Assume $$R = 8.0$$ J mol$$^{-1}$$ K$$^{-1}$$. | (Q) 7 kJ |
| (III) One mole of a monatomic ideal gas is compressed adiabatically from volume $$V = \frac{1}{3}$$ m$$^3$$ and pressure 2 kPa to volume $$\frac{V}{8}$$. | (R) 4 kJ |
| (IV) Three moles of a diatomic ideal gas whose molecules can vibrate, is given 9 kJ of heat and undergoes isobaric expansion. | (S) 5 kJ |
| (T) 3 kJ |
Which one of the following options is correct?
JEE Advanced 2022 Paper-1 - Question 36
List I contains four combinations of two lenses (1 and 2) whose focal lengths (in cm) are indicated in the figures. In all cases, the object is placed 20 cm from the first lens on the left, and the distance between the two lenses is 5 cm. List II contains the positions of the final images.
| List-I | List-II |
|---|---|
(I) 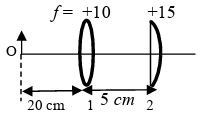 | (P) Final image is formed at 7.5 cm on the right side of lens 2. |
(II)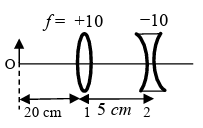 | (Q) Final image is formed at 60.0 cm on the right side of lens 2. |
(III) 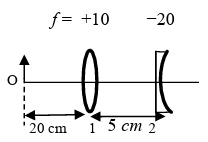 | (R) Final image is formed at 30.0 cm on the left side of lens 2. |
(IV) 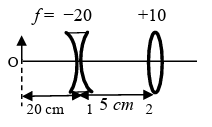 | (S) Final image is formed at 6.0 cm on the right side of lens 2. |
| (T) Final image is formed at 30.0 cm on the right side of lens 2. |
Which one of the following options is correct?
JEE Advanced 2022 Paper-1 - Question 37
2 mol of Hg(g) is combusted in a fixed volume bomb calorimeter with excess of $$O_2$$ at 298 K and 1 atm into HgO(s). During the reaction, temperature increases from 298.0 K to 312.8 K. If heat capacity of the bomb calorimeter and enthalpy of formation of Hg(g) are 20.00 kJ K$$^{-1}$$ and 61.32 kJ mol$$^{-1}$$ at 298 K, respectively, the calculated standard molar enthalpy of formation of HgO(s) at 298 K is X kJ mol$$^{-1}$$. The value of |X| is ______.
[Given : Gas constant R = 8.3 J K$$^{-1}$$ mol$$^{-1}$$]
789
456
123
0.-
Clear All
JEE Advanced 2022 Paper-1 - Question 38
The reduction potential ($$E^0$$, in V) of $$MnO_4^-$$(aq)/Mn(s) is ______.
[Given : $$E^0_{MnO_4^-(aq)/MnO_2(s)} = 1.68$$ V ; $$E^0_{MnO_2(s)/Mn^{2+}(aq)} = 1.21$$ V ; $$E^0_{Mn^{2+}(aq)/Mn(s)} = -1.03$$ V]
789
456
123
0.-
Clear All
JEE Advanced 2022 Paper-1 - Question 39
A solution is prepared by mixing 0.01 mol each of $$H_2CO_3$$, NaHCO$$_3$$, Na$$_2$$CO$$_3$$, and NaOH in 100 mL of water. pH of the resulting solution is ______.
[Given : $$pK_{a1}$$ and $$pK_{a2}$$ of $$H_2CO_3$$ are 6.37 and 10.32, respectively ; log 2 = 0.30]
789
456
123
0.-
Clear All
JEE Advanced 2022 Paper-1 - Question 40
The treatment of an aqueous solution of 3.74 g of Cu(NO$$_3$$)$$_2$$ with excess KI results in a brown solution along with the formation of a precipitate. Passing H$$_2$$S through this brown solution gives another precipitate X. The amount of X (in g) is ______.
[Given : Atomic mass of H = 1, N = 14, O = 16, S = 32, K = 39, Cu = 63, I = 127]
789
456
123
0.-
Clear All



