JEE Advanced 2022 Paper-1
For the following questions answer them individually
JEE Advanced 2022 Paper-1 - Question 41
Dissolving 1.24 g of white phosphorous in boiling NaOH solution in an inert atmosphere gives a gas Q. The amount of CuSO$$_4$$ (in g) required to completely consume the gas Q is ______.
[Given : Atomic mass of H = 1, O = 16, Na = 23, P = 31, S = 32, Cu = 63]
789
456
123
0.-
Clear All
JEE Advanced 2022 Paper-1 - Question 42
Consider the following reaction.
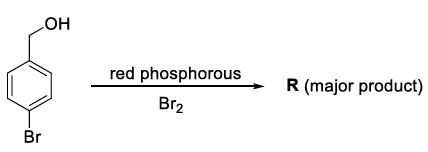
On estimation of bromine in 1.00 g of R using Carius method, the amount of AgBr formed (in g) is ______.
[Given : Atomic mass of H = 1, C = 12, O = 16, P = 31, Br = 80, Ag = 108]
789
456
123
0.-
Clear All
JEE Advanced 2022 Paper-1 - Question 43
The weight percentage of hydrogen in Q, formed in the following reaction sequence, is ______.
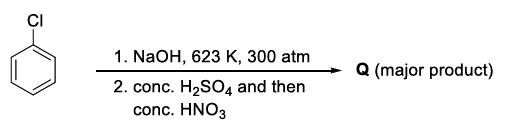
[Given : Atomic mass of H = 1, C = 12, N = 14, O = 16, S = 32, Cl = 35]
789
456
123
0.-
Clear All
JEE Advanced 2022 Paper-1 - Question 44
If the reaction sequence given below is carried out with 15 moles of acetylene, the amount of product D formed (in g) is ______.
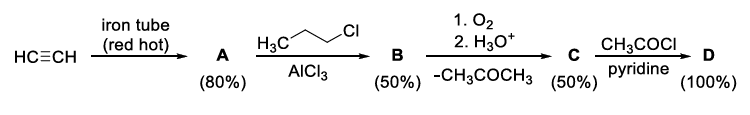
The yields of A, B, C and D are given in parentheses.
[Given : Atomic mass of H = 1, C = 12, O = 16, Cl = 35]
789
456
123
0.-
Clear All
JEE Advanced 2022 Paper-1 - Question 45
For diatomic molecules, the correct statement(s) about the molecular orbitals formed by the overlap of two 2$$p_z$$ orbitals is(are)
JEE Advanced 2022 Paper-1 - Question 46
The correct option(s) related to adsorption processes is(are)
JEE Advanced 2022 Paper-1 - Question 47
The electrochemical extraction of aluminum from bauxite ore involves
JEE Advanced 2022 Paper-1 - Question 48
The treatment of galena with HNO$$_3$$ produces a gas that is
JEE Advanced 2022 Paper-1 - Question 49
Considering the reaction sequence given below, the correct statement(s) is(are)
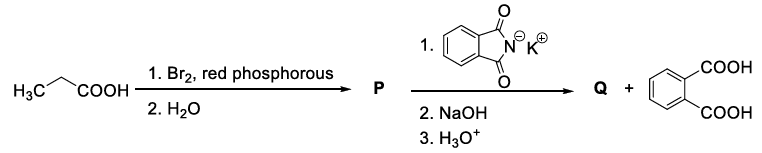
JEE Advanced 2022 Paper-1 - Question 50
Consider the following reaction sequence,
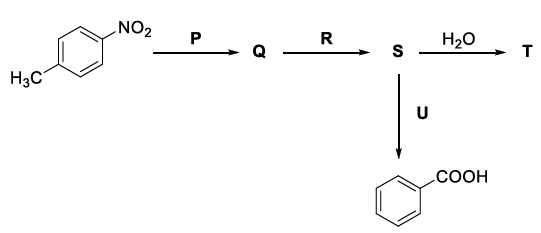
the correct option(s) is(are)



