Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
For the reaction, $$aA + bB \to cC + dD$$, the plot of log k v/s $$\frac{1}{T}$$ is given below:
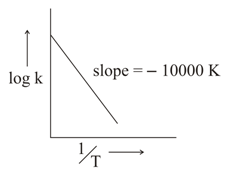
The temperature at which the rate constant of the reaction is $$10^{-4}$$ s$$^{-1}$$ is ______ K.
(Rounded-off to the nearest integer) [Given: The rate constant of the reaction is $$10^{-5}$$ s$$^{-1}$$ at 500 K.]
Correct Answer: 526
The Arrhenius equation is:
$$\mathrm{\log k = \log A - \frac{E_a}{2.303RT}}$$
Comparing with the straight-line equation:
$$\mathrm{y = mx + c}$$
for a plot of $$\mathrm{\log k}$$ versus $$\mathrm{\frac{1}{T}}$$:
$$\mathrm{Slope = -\frac{E_a}{2.303R}}$$
Given slope:
$$\mathrm{m = -10000\ K}$$
Using the two-point equation:
$$\mathrm{\log k_2 - \log k_1 = m\left(\frac{1}{T_2}-\frac{1}{T_1}\right)}$$
Given:
$$\mathrm{k_1 = 10^{-5}\ s^{-1}}$$
$$\mathrm{k_2 = 10^{-4}\ s^{-1}}$$
$$\mathrm{T_1 = 500\ K}$$
Substituting values:
$$\mathrm{\log(10^{-4}) - \log(10^{-5}) = -10000\left(\frac{1}{T_2}-\frac{1}{500}\right)}$$
$$\mathrm{-4 - (-5) = -10000\left(\frac{1}{T_2}-\frac{1}{500}\right)}$$
$$\mathrm{1 = -10000\left(\frac{1}{T_2}-\frac{1}{500}\right)}$$
$$\mathrm{-\frac{1}{10000} = \frac{1}{T_2}-\frac{1}{500}}$$
$$\mathrm{\frac{1}{T_2} = \frac{1}{500}-\frac{1}{10000}}$$
Taking LCM:
$$\mathrm{\frac{1}{T_2} = \frac{20}{10000}-\frac{1}{10000}}$$
$$\mathrm{\frac{1}{T_2} = \frac{19}{10000}}$$
$$\mathrm{T_2 = \frac{10000}{19}}$$
$$\mathrm{T_2 \approx 526.31\ K}$$
Rounding to nearest integer:
$$\boxed{\mathrm{526}}$$
Create a FREE account and get:
Educational materials for JEE preparation
