Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Consider the following reaction sequence
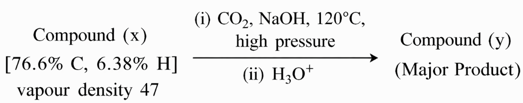
Compound (y) develops characterstic colour with neutral $$FeCl_{3}$$ solution.
Identify the INCORRECT statement from the following for the above sequence.
To find the molecular formula of x, we use the percentage composition and vapor density:
Phenol undergoes the Kolbe-Schmitt Reaction when treated with $$CO_2$$ and $$NaOH$$ under high pressure, followed by acidification.
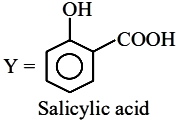
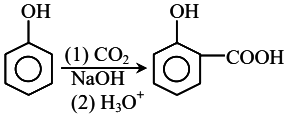
Based on the properties of Phenol (x) and Salicylic acid (y):
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
