Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
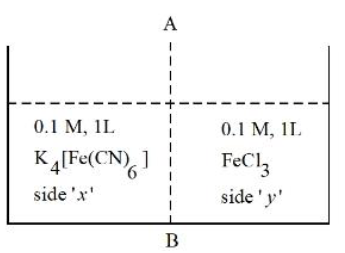
At 27 °C, 0.1 M, 1 L K$$_4$$[Fe(CN)$$_6$$] aqueous solution and 0.1 M, 1 L FeCl$$_3$$ aqueous solution are placed in a container separated by a semi permeable membrane AB. Assume complete dissociation of both the solutes. Which of the following statement is correct?
Osmotic pressure of a dilute solution is given by the van’t Hoff relation
$$\Pi = i\,C\,R\,T$$
where $$i$$ is the van’t Hoff factor (total ions produced per formula unit), $$C$$ is the molarity, $$R$$ is the gas constant and $$T$$ is the absolute temperature. At the same temperature and with equal volumes, the solution having the larger value of $$i\,C$$ possesses the larger osmotic pressure and is called hypertonic; the other one is hypotonic.
Step 1 : Dissociation and van’t Hoff factors
For $$K_4[Fe(CN)_6]$$: $$K_4[Fe(CN)_6] \rightarrow 4\,K^{+} + [Fe(CN)_6]^{4-}$$
Total ions = $$4 + 1 = 5 \;\Rightarrow\; i_x = 5$$
For $$FeCl_3$$: $$FeCl_3 \rightarrow Fe^{3+} + 3\,Cl^{-}$$
Total ions = $$1 + 3 = 4 \;\Rightarrow\; i_y = 4$$
Step 2 : Particle (osmotic) concentrations
Both solutions have the same molarity $$C = 0.1\,$$M.
Side x (containing $$K_4[Fe(CN)_6]$$): $$i\,C = 5 \times 0.1 = 0.50\;\text{mol L}^{-1}$$
Side y (containing $$FeCl_3$$): $$i\,C = 4 \times 0.1 = 0.40\;\text{mol L}^{-1}$$
Step 3 : Comparison of osmotic pressures
Because $$\Pi \propto i\,C$$ at the same $$T$$,
$$\Pi_x = 0.50\,R\,T \gt \Pi_y = 0.40\,R\,T$$
Therefore side x is hypertonic (higher osmotic pressure) and side y is hypotonic (lower osmotic pressure).
Step 4 : Verification of the given options
A. Prussian blue forms only if $$Fe^{3+}$$ meets $$[Fe(CN)_6]^{4-}$$. The semipermeable membrane blocks solute ions, so no mixing, no precipitate.
B. By definition, ionic solutes cannot pass through a semipermeable (osmotic) membrane — only the solvent moves.
C. As shown above, the $$FeCl_3$$ side (solution y) has the lower osmotic pressure, hence it is hypotonic — this statement is correct.
D. Reverse osmosis would require pressure on the hypertonic side (side x). Whether this matches the wording depends on the diagram, but only Option C is unambiguously correct from the data given.
Hence, the correct choice is:
Option C which is: Solution y is hypotonic.
Create a FREE account and get:
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation
