Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
$$A(g) \to B(g) + C(g)$$ is a first order reaction.
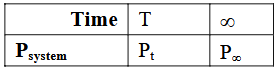
The reaction was started with reactant A only. Which of the following expression is correct for rate constant k?
For the gas-phase decomposition $$A(g) \rightarrow B(g)+C(g)$$ at constant temperature and volume, the total pressure is directly proportional to the total number of moles present at any instant.
Let the reaction start with $$a$$ moles of $$A$$ and no $$B$$ or $$C$$.
At time $$t$$, let $$x$$ moles of $$A$$ decompose.
Moles present at time $$t$$:
$$A : a-x$$
$$B : x$$
$$C : x$$
Total moles at time $$t$$ are $$n_t = a-x + x + x = a + x$$.
If $$f = \dfrac{RT}{V}$$ (a common proportionality factor), then
initial pressure, $$P_0 = f a$$,
pressure at time $$t$$, $$P_t = f (a + x) = P_0 + f x$$ $$-(1)$$
From $$(1)$$, $$x = \dfrac{P_t - P_0}{f}$$ $$-(2)$$
When the reaction goes to completion ($$t \rightarrow \infty$$), $$x = a$$.
Total moles then are $$a + a = 2a$$, so the final pressure is
$$P_{\infty} = f(2a) = 2 P_0$$ $$\Longrightarrow P_0 = \dfrac{P_{\infty}}{2}$$ $$-(3)$$
The first-order rate law is $$k = \dfrac{1}{t} \ln \dfrac{\text{initial concentration of }A}{\text{concentration of }A\text{ at time }t}$$, i.e.
$$k = \dfrac{1}{t} \ln \dfrac{a}{a - x}$$ $$-(4)$$
Using $$(2)$$, $$a - x = \dfrac{P_0}{f} - \dfrac{P_t - P_0}{f} = \dfrac{2P_0 - P_t}{f}$$.
Thus,
$$\dfrac{a}{a - x} = \dfrac{\dfrac{P_0}{f}}{\dfrac{2P_0 - P_t}{f}} = \dfrac{P_0}{2P_0 - P_t}$$ $$-(5)$$
Substitute $$P_0 = \dfrac{P_{\infty}}{2}$$ from $$(3)$$ into $$(5)$$:
$$\dfrac{a}{a - x} = \dfrac{\dfrac{P_{\infty}}{2}}{P_{\infty} - P_t} = \dfrac{P_{\infty}}{2(P_{\infty} - P_t)}$$ $$-(6)$$
Insert $$(6)$$ into the rate law $$(4)$$:
$$k = \dfrac{1}{t} \ln \left[ \dfrac{P_{\infty}}{2(P_{\infty} - P_t)} \right]$$
This matches Option C.
Hence, the correct expression for the first-order rate constant is
Case C: $$\;k = \dfrac{1}{t} \ln \dfrac{p_{\infty}}{2(p_{\infty} - P_t)}$$.
Create a FREE account and get:
Educational materials for JEE preparation
