Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Match the LIST-I with LIST-II.
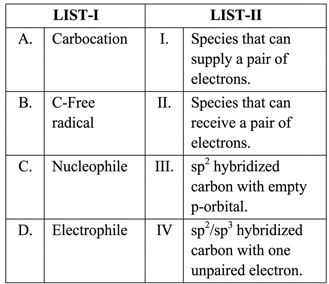
Choose the correct answer from the options given below :
For every species, identify its general electronic feature and hybridisation before matching with the statements in LIST-II.
Case A: Carbocation
A carbocation carries a positive charge on carbon. The carbon is $$sp^2$$ hybridised and possesses an empty $$p$$-orbital that can accept a pair of electrons.
Hence, A corresponds to statement III.
Case B: Carbon-centred free radical
A carbon free radical has one unpaired electron on carbon. Depending on the surrounding atoms it may be $$sp^2$$ or $$sp^3$$ hybridised, but the defining feature is the single unpaired electron.
Hence, B corresponds to statement IV.
Case C: Nucleophile
A nucleophile is a species that donates (supplies) an electron pair to an electron-deficient centre in a reaction.
Hence, C corresponds to statement I.
Case D: Electrophile
An electrophile is electron-deficient; it seeks (receives) an electron pair from a nucleophile.
Hence, D corresponds to statement II.
Collecting the matches:
A - III, B - IV, C - I, D - II.
This is exactly the pairing shown in Option D. Therefore the correct answer is Option D.
Create a FREE account and get:
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation
