Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
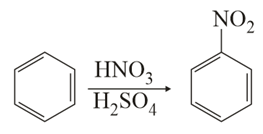
In the above reaction, 3.9 g of benzene on nitration gives 4.92 g of nitrobenzene. The percentage yield of nitrobenzene in the above reaction is ________ %. (Round off to the Nearest Integer).
(Given atomic mass: C: 12.0 u, H: 1.0 u, O: 16.0 u, N: 14.0 u)
Correct Answer: 80
The nitration of benzene produces nitrobenzene: $$C_6H_6 + HNO_3 \xrightarrow{H_2SO_4} C_6H_5NO_2 + H_2O$$. The molar mass of benzene ($$C_6H_6$$) is $$6 \times 12 + 6 \times 1 = 78$$ g/mol, and the molar mass of nitrobenzene ($$C_6H_5NO_2$$) is $$6 \times 12 + 5 \times 1 + 14 + 2 \times 16 = 123$$ g/mol.
From 3.9 g of benzene, the number of moles is $$\frac{3.9}{78} = 0.05$$ mol. Since the reaction has a 1:1 stoichiometry, the theoretical yield of nitrobenzene is $$0.05 \times 123 = 6.15$$ g.
The actual yield is 4.92 g. Therefore, the percentage yield is $$\frac{4.92}{6.15} \times 100 = 80\%$$.
Create a FREE account and get:
Educational materials for JEE preparation
