Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
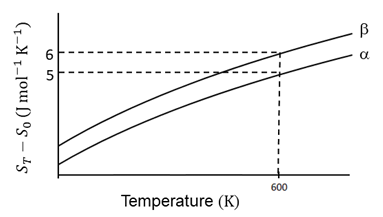
The entropy versus temperature plot for phases $$\alpha$$ and $$\beta$$ at 1 bar pressure is given. $$S_T$$ and $$S_0$$ are entropies of the phases at temperatures T and 0 K, respectively.
The transition temperature for $$\alpha$$ to $$\beta$$ phase change is 600 K and $$C_{P,\beta} - C_{P,\alpha} = 1$$ J mol$$^{-1}$$ K$$^{-1}$$. Assume $$(C_{P,\beta} - C_{P,\alpha})$$ is independent of temperature in the range of 200 to 700 K. $$C_{P,\alpha}$$ and $$C_{P,\beta}$$ are heat capacities of $$\alpha$$ and $$\beta$$ phases, respectively.
The value of enthalpy change, $$H_\beta - H_\alpha$$ (in J mol$$^{-1}$$), at 300 K is ____.
Correct Answer: 300
The enthalpy difference between the two phases at any temperature $$T$$ can be obtained by integrating the heat-capacity difference from 0 K to $$T$$.
For the given data, the heat-capacity difference is constant:
$$C_{P,\beta}-C_{P,\alpha}=1\ \text{J mol}^{-1}\text{K}^{-1}$$
Starting from 0 K (where both phases may be taken to have the same enthalpy), the enthalpy difference at temperature $$T$$ is
$$\begin{aligned} H_\beta-H_\alpha &= \int_{0}^{T}\bigl(C_{P,\beta}-C_{P,\alpha}\bigr)\,dT \\ &= (C_{P,\beta}-C_{P,\alpha})\int_{0}^{T} dT \\ &= (C_{P,\beta}-C_{P,\alpha})\,T \end{aligned}$$
Substituting the numerical values for the required temperature $$T=300\ \text{K}$$:
$$H_\beta-H_\alpha = 1\ \text{J mol}^{-1}\text{K}^{-1}\times 300\ \text{K} = 300\ \text{J mol}^{-1}$$
Hence, the enthalpy change at 300 K is 300 J mol$$^{-1}$$.
Create a FREE account and get:
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation
