Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
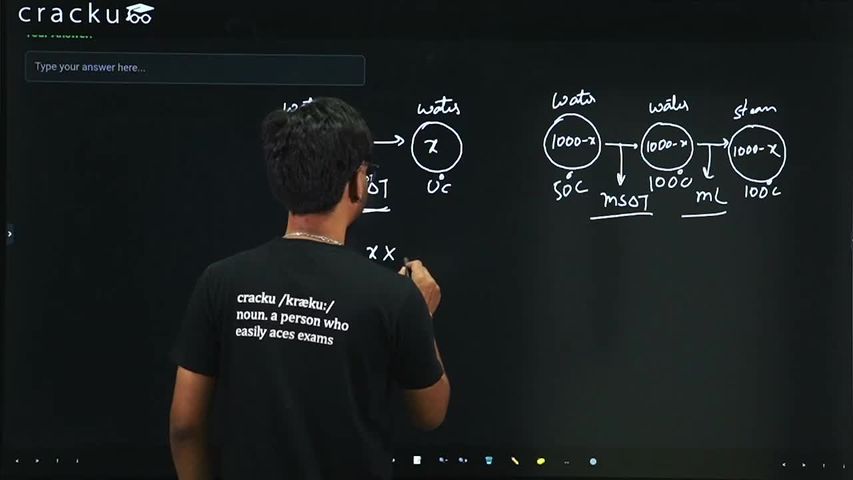
The heat extracted out of x gram of water initialy at $$50°$$C to cool it down to $$0°$$C is sufficient to evaporate $$(1000 - x)$$ gram of water also initialy at $$50°$$C. The value of $$x$$ (closest integer) is_______. (Take Latent heat of water $$L = 2256$$ kJ/kg K, specific heat capacity of water $$c = 4200$$ J/kg·K)
Correct Answer: 922
$$\text{Heat Lost} = \text{Heat Gained}$$
$$Q = mc\Delta T$$ (temperature change without phase change)
$$Q = mL$$ (Phase change)
The first part of the water ($$x$$ grams) is cooled from $$50^\circ\text{C}$$ to $$0^\circ\text{C}$$.
$$Q_{\text{extracted}} = m_1 \cdot c \cdot \Delta T$$
$$Q_{\text{extracted}} = \left(x \times 10^{-3}\right) \times 4200 \times 50$$
$$Q_{\text{extracted}} = x \times 10^{-3} \times 210,000 = 210x\text{ J}$$
The remaining water, $$(1000-x)$$ grams, is initially at $$50^\circ\text{C}$$. To evaporate it, it must first be heated to its boiling point ($$100^\circ\text{C}$$) and then converted into steam at $$100^\circ\text{C}$$.
The total heat required ($$Q_{\text{required}}$$) consists of two parts:
Heating from $$50^\circ\text{C}$$ to $$100^\circ\text{C}$$ ($$\Delta T_2 = 100 - 50 = 50\text{ K}$$): $$Q_{\text{heating}} = m_2 \cdot c \cdot \Delta T_2$$
Evaporating at $$100^\circ\text{C}$$: $$Q_{\text{phase\_change}} = m_2 \cdot L$$
$$Q_{\text{required}} = m_2 \left(c\Delta T_2 + L\right)$$
$$Q_{\text{required}} = (1000 - x) \times 10^{-3} \times \left[(4200 \times 50) + \left(2256 \times 10^3\right)\right]$$
$$Q_{\text{required}} = 2466(1000 - x)\text{ J}$$
$$Q_{\text{extracted}} = Q_{\text{required}}$$
$$210x = 2466(1000 - x)$$
$$x = \frac{2,466,000}{2676}$$
$$x \approx 921.52 \approx 922$$
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
