Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Following figure shows dependence of molar conductance of two electrolytes on concentration. $$\Lambda^0_m$$ is the limiting molar conductivity.
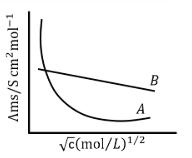
The number of Incorrect statement(s) from the following is ______
(A) $$\Lambda^0_m$$ for electrolyte A is obtained by extrapolation
(B) For electrolyte B, $$\Lambda_m$$ Vs $$\sqrt{c}$$ graph is a straight line with intercept equal to $$\Lambda^0_m$$
(C) At infinite dilution, the value of degree of dissociation approach zero for electrolyte B.
(D) $$\Lambda_m$$ for any electrolyte A or B can be calculated using $$\lambda°$$ for individual ions.
Correct Answer: 3
Create a FREE account and get:
Educational materials for JEE preparation
