Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
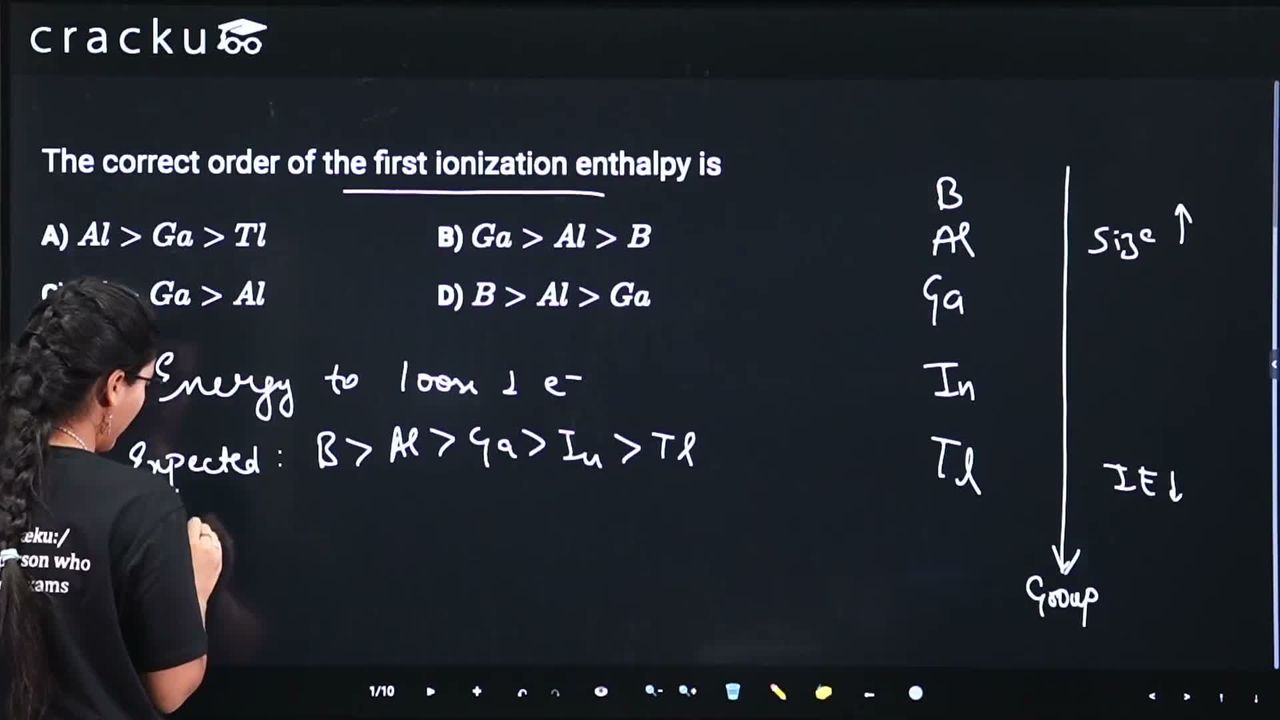
We need to find the correct order of first ionization enthalpy for Group 13 elements.
The first ionization enthalpies (in kJ/mol, approximate) for Group 13 elements are:
B: 801, Al: 577, Ga: 579, In: 558, Tl: 589
The general trend down a group is decreasing IE due to increasing atomic size and shielding. However, anomalies exist:
Ga vs Al: Ga (579) has slightly higher IE than Al (577) due to poor shielding by the 3d electrons in Ga, leading to higher effective nuclear charge.
Tl vs Ga: Tl (589) has higher IE than Ga (579) due to the poor shielding effect of 4f electrons (lanthanide contraction) and relativistic effects on the 6s electrons.
Among the given options, the order $$Tl > Ga > Al$$ (589 > 579 > 577) is correct.
The correct answer is Option C: $$Tl > Ga > Al$$.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
