Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
It is observed that characteristic X-ray spectra of elements show regularity. When frequency to the power 'n' i.e. $$\nu^n$$ of X-rays emitted is plotted against atomic number Z, the following graph is obtained.
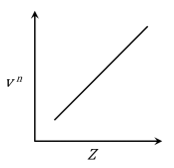
The value of 'n' is
According to Moseley's Law for characteristic X-rays:
$$\sqrt{\nu} = a(Z - b)$$
Where $a$ and $b$ are constants.
For the graph to be a straight line (linear) when plotted against $Z$:
$$\nu^n \propto Z$$
Rewriting Moseley's Law:
$$\nu^{1/2} = aZ - ab$$
Comparing the power of $\nu$ in the equation to the power $n$ in the graph:
$$n = \frac{1}{2}$$
$$\boxed{n = \frac{1}{2}}$$
Create a FREE account and get:
Educational materials for JEE preparation
