Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Identify the elements X and Y using the ionization energy values given below:
Ionization energy (kJ/mol):
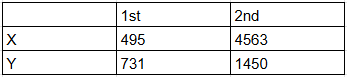
Element X has a 1st ionization energy of 495 kJ/mol and a very high 2nd ionization energy of 4563 kJ/mol. The huge jump between the 1st and 2nd ionization energies indicates that X has only 1 valence electron. This is characteristic of sodium (Na), which has the configuration $$[Ne]3s^1$$. After removing the single valence electron, the next electron must come from the stable noble gas core, requiring much more energy.
Element Y has a 1st ionization energy of 731 kJ/mol and a 2nd ionization energy of 1450 kJ/mol. The ratio of the 2nd to 1st IE is roughly 2, with no dramatic jump. This suggests Y has more than one valence electron. Magnesium (Mg) with configuration $$[Ne]3s^2$$ has two valence electrons and shows a gradual increase in successive ionization energies, consistent with these values.
Therefore, X = Na and Y = Mg, which corresponds to option (1).
Create a FREE account and get:
Educational materials for JEE preparation
