Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
An ideal monatomic gas of $$n$$ moles is taken through a cycle $$WXYZW$$ consisting of consecutive adiabatic and isobaric quasi-static processes, as shown in the schematic $$V$$-$$T$$ diagram. The volume of the gas at $$W$$, $$X$$ and $$Y$$ points are, 64 cm$$^3$$, 125 cm$$^3$$ and 250 cm$$^3$$, respectively. If the absolute temperature of the gas $$T_W$$ at the point $$W$$ is such that $$nRT_W = 1$$ J ($$R$$ is the universal gas constant), then the amount of heat absorbed (in J) by the gas along the path $$XY$$ is ______.
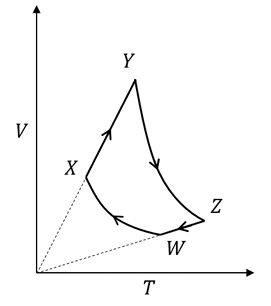
Correct Answer: 1.6
For an ideal monatomic gas, the ratio of specific heats is $$\gamma = \dfrac{5}{3}$$.
Case 1: Adiabatic branch $$W \rightarrow X$$
For a reversible adiabatic process,
$$T V^{\,\gamma-1} = \text{constant}$$
Thus $$T_W V_W^{\,\gamma-1} = T_X V_X^{\,\gamma-1}$$ $$$ \Rightarrow \; T_X = T_W \left(\dfrac{V_W}{V_X}\right)^{\gamma-1} = T_W \left(\dfrac{64}{125}\right)^{\frac{2}{3}} $$$
Since $$64 = 4^3$$ and $$125 = 5^3$$, $$\left(\dfrac{64}{125}\right)^{\frac{2}{3}} = \left(\dfrac{4^3}{5^3}\right)^{\frac{2}{3}} = \left(\dfrac{4}{5}\right)^{2} = \dfrac{16}{25} = 0.64$$
Therefore, $$T_X = 0.64 \,T_W \qquad -(1)$$
Case 2: Isobaric branch $$X \rightarrow Y$$
For an isobaric process, $$P$$ is constant, so from the ideal-gas law $$PV = nRT$$ we have
$$\dfrac{T}{V} = \text{constant} \;\Longrightarrow\; \dfrac{T_Y}{T_X} = \dfrac{V_Y}{V_X}$$
Given $$V_Y = 250\;\text{cm}^3$$ and $$V_X = 125\;\text{cm}^3$$, $$\dfrac{T_Y}{T_X} = \dfrac{250}{125} = 2 \;\Longrightarrow\; T_Y = 2\,T_X \qquad -(2)$$
Using $$(1)$$ in $$(2)$$, $$T_Y = 2 \times 0.64\,T_W = 1.28\,T_W$$
Hence the temperature rise along $$X \rightarrow Y$$ is $$\Delta T = T_Y - T_X = 1.28\,T_W - 0.64\,T_W = 0.64\,T_W$$
The heat absorbed in an isobaric process is $$Q_{XY} = n C_p \Delta T$$ where, for a monatomic gas, $$C_p = \dfrac{5}{2}R$$.
Therefore, $$Q_{XY} = n\left(\dfrac{5}{2}R\right)(0.64\,T_W) = \dfrac{5}{2} \times 0.64 \; (nRT_W)$$
The problem states that $$nRT_W = 1\;\text{J}$$, so $$Q_{XY} = \dfrac{5}{2} \times 0.64 \times 1 = 2.5 \times 0.64 = 1.6\;\text{J}$$
Hence, the heat absorbed by the gas along the path $$X \rightarrow Y$$ is 1.6 J.
Create a FREE account and get:
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation
