Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
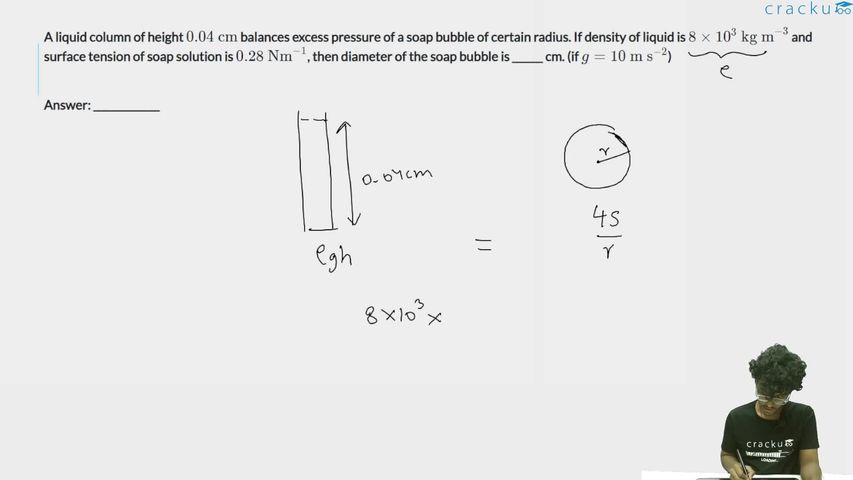
A liquid column of height $$0.04 \text{ cm}$$ balances excess pressure of a soap bubble of certain radius. If density of liquid is $$8 \times 10^3 \text{ kg m}^{-3}$$ and surface tension of soap solution is $$0.28 \text{ Nm}^{-1}$$, then diameter of the soap bubble is ______ cm. (if $$g = 10 \text{ m s}^{-2}$$)
Correct Answer: 7
A liquid column of height 0.04 cm balances the excess pressure of a soap bubble. Find the diameter of the bubble.
$$\Delta P = \frac{4S}{R}$$
where $$S$$ is the surface tension and $$R$$ is the radius. (Factor of 4 because a soap bubble has two surfaces.)
$$\Delta P = \rho g h = 8000 \times 10 \times 0.04 \times 10^{-2} = 8000 \times 10 \times 4 \times 10^{-4} = 32 \text{ Pa}$$
$$\frac{4S}{R} = \rho g h \implies R = \frac{4S}{\rho g h} = \frac{4 \times 0.28}{32} = \frac{1.12}{32} = 0.035 \text{ m}$$
$$D = 2R = 0.07 \text{ m} = 7 \text{ cm}$$
The correct answer is 7.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
