Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
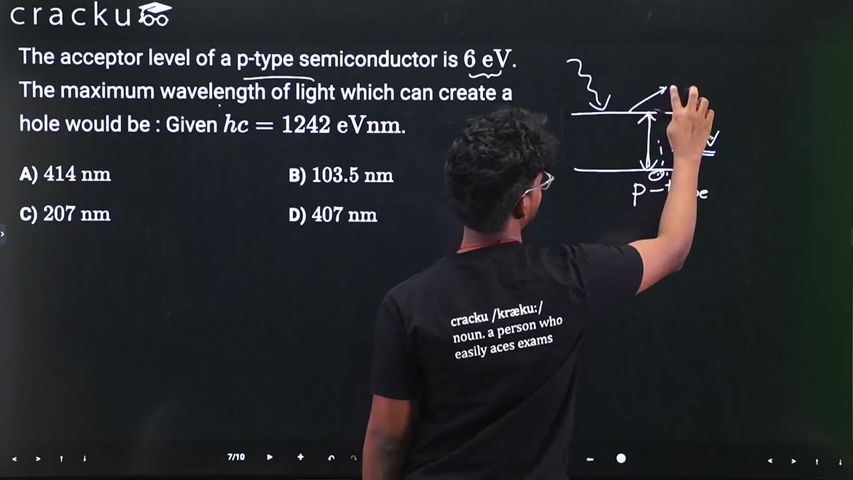
The acceptor level of a p-type semiconductor is $$6 \text{ eV}$$. The maximum wavelength of light which can create a hole would be : Given $$hc = 1242 \text{ eVnm}$$.
To create a hole in a p-type semiconductor, an electron must be excited from the valence band to the acceptor energy level. The minimum energy required for this transition is the energy gap of the acceptor level.
$$ E = 6 \text{ eV} $$
$$ E = \frac{hc}{\lambda} $$
$$ \lambda_{max} = \frac{hc}{E} $$
$$ hc = 1242 \text{ eVnm} $$
$$ E = 6 \text{ eV} $$
$$ \lambda_{max} = \frac{1242}{6} $$
$$ \lambda_{max} = 207 \text{ nm} $$
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
